The Acid Sea
The carbon dioxide we pump into the air is seeping into the oceans and slowly acidifying them. One hundred years from now, will oysters, mussels, and coral reefs survive?
Castello Aragonese is a tiny island that rises straight out of the Tyrrhenian Sea like a tower. Seventeen miles west of Naples, it can be reached from the somewhat larger island of Ischia via a long, narrow stone bridge. The tourists who visit Castello Aragonese come to see what life was like in the past. They climb—or better yet, take the elevator—up to a massive castle, which houses a display of medieval torture instruments. The scientists who visit the island, by contrast, come to see what life will be like in the future.
Owing to a quirk of geology, the sea around Castello Aragonese provides a window onto the oceans of 2050 and beyond. Bubbles of CO2 rise from volcanic vents on the seafloor and dissolve to form carbonic acid. Carbonic acid is relatively weak; people drink it all the time in carbonated beverages. But if enough of it forms, it makes seawater corrosive. "When you get to the extremely high CO2, almost nothing can tolerate that," Jason Hall-Spencer, a marine biologist from Britain's University of Plymouth, explains. Castello Aragonese offers a natural analogue for an unnatural process: The acidification that has taken place off its shore is occurring more gradually across the world's oceans, as they absorb more and more of the carbon dioxide that's coming from tailpipes and smokestacks.
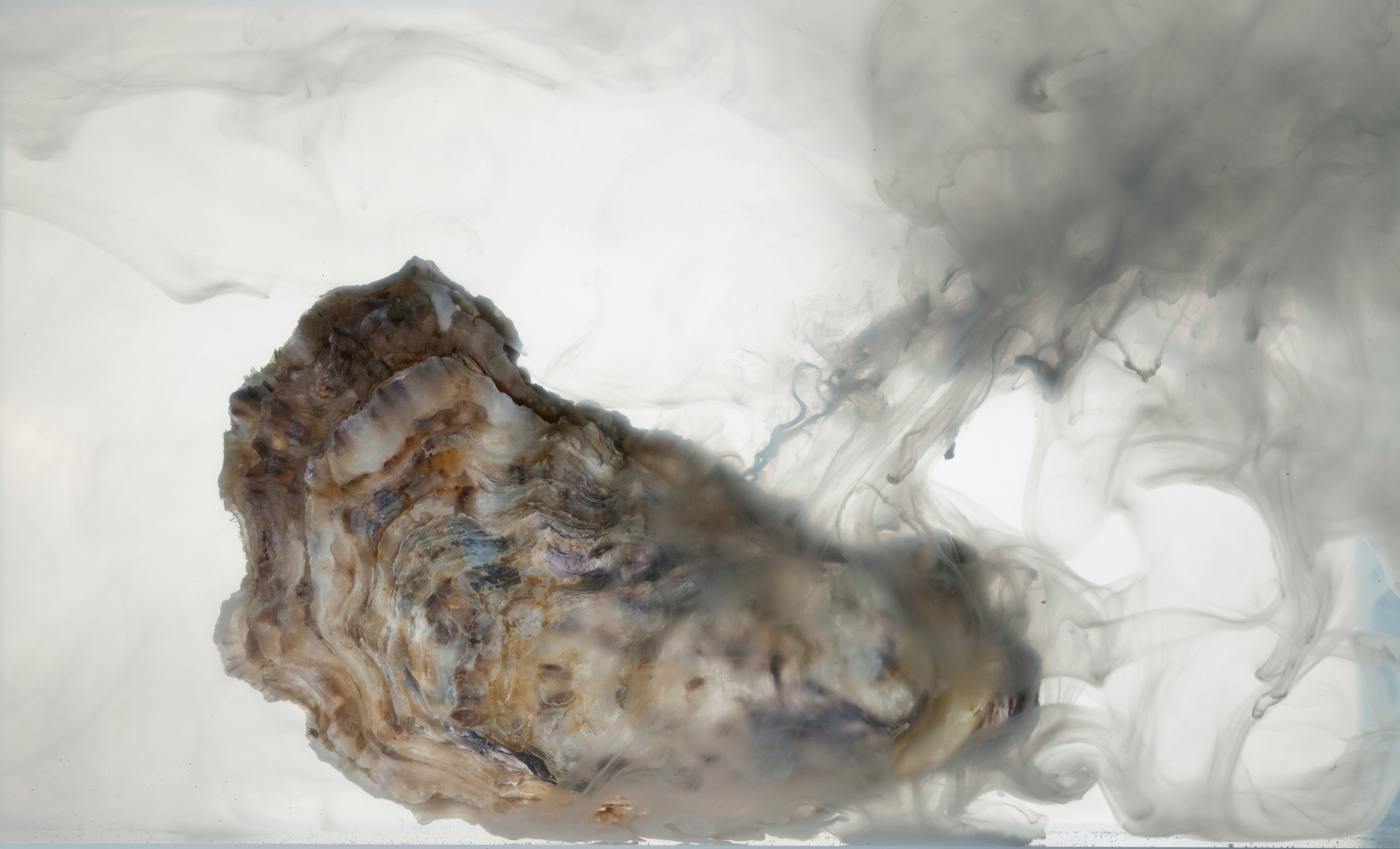
Hall-Spencer has been studying the sea around the island for the past eight years, carefully measuring the properties of the water and tracking the fish and corals and mollusks that live and, in some cases, dissolve there. On a chilly winter's day I went swimming with him and with Maria Cristina Buia, a scientist at Italy's Anton Dohrn Zoological Station, to see the effects of acidification up close. We anchored our boat about 50 yards from the southern shore of Castello Aragonese. Even before we got into the water, some impacts were evident. Clumps of barnacles formed a whitish band at the base of the island's wave-battered cliffs. "Barnacles are really tough," Hall-Spencer observed. In the areas where the water was most acidified, though, they were missing.
We all dived in. Buia was carrying a knife. She pried some unlucky limpets from a rock. Searching for food, they had wandered into water that was too caustic for them. Their shells were so thin they were almost transparent. Bubbles of carbon dioxide streamed up from the seafloor like beads of quicksilver. We swam on. Beds of sea grass waved beneath us. The grass was a vivid green; the tiny organisms that usually coat the blades, dulling their color, were all missing. Sea urchins, commonplace away from the vents, were also absent; they can't tolerate even moderately acidified water. Swarms of nearly transparent jellyfish floated by. "Watch out," Hall-Spencer warned. "They sting."
Jellyfish, sea grass, and algae—not much else lives near the densest concentration of vents at Castello Aragonese. Even a few hundred yards away, many native species can't survive. The water there is about as acidified as the oceans as a whole are forecast to be by 2100. "Normally in a polluted harbor you've got just a few species that are weedlike and able to cope with widely fluctuating conditions," Hall-Spencer said once we were back on the boat. "Well, it's like that when you ramp up CO2."
Since the start of the industrial revolution, enough fossil fuels—coal, oil, and natural gas—have been burned and enough forests cut down to emit more than 500 billion tons of CO2. As is well known, the atmosphere has a higher concentration of CO2today than at any point in the past 800,000 years and probably a lot longer.
What is less well known is how carbon emissions are changing the oceans too. The air and the water constantly exchange gases, so a portion of anything emitted into the atmosphere eventually ends up in the sea. Winds quickly mix it into the top few hundred feet, and over centuries currents spread it through the ocean depths. In the 1990s an international team of scientists undertook a massive research project that involved collecting and analyzing more than 77,000 seawater samples from different depths and locations around the world. The work took 15 years. It showed that the oceans have absorbed 30 percent of the CO2 released by humans over the past two centuries. They continue to absorb roughly a million tons every hour.
For life on land this process is a boon; every ton of CO2 the oceans remove from the atmosphere is a ton that's not contributing to global warming. But for life in the sea the picture looks different. The head of the National Oceanic and Atmospheric Administration, Jane Lubchenco, a marine ecologist, has called ocean acidification global warming's "equally evil twin."
The pH scale, which measures acidity in terms of the concentration of hydrogen ions, runs from zero to 14. At the low end of the scale are strong acids, such as hydrochloric acid, that release hydrogen readily (more readily than carbonic acid does). At the high end are strong bases such as lye. Pure, distilled water has a pH of 7, which is neutral. Seawater should be slightly basic, with a pH around 8.2 near the sea surface. So far CO2 emissions have reduced the pH there by about 0.1. Like the Richter scale, the pH scale is logarithmic, so even small numerical changes represent large effects. A pH drop of 0.1 means the water has become 30 percent more acidic. If present trends continue, surface pH will drop to around 7.8 by 2100. At that point the water will be 150 percent more acidic than it was in 1800.
The acidification that has occurred so far is probably irreversible. Although in theory it's possible to add chemicals to the sea to counter the effects of the extra CO2, as a practical matter, the volumes involved would be staggering; it would take at least two tons of lime, for example, to offset a single ton of carbon dioxide, and the world now emits more than 30 billion tons of CO2 each year. Meanwhile, natural processes that could counter acidification—such as the weathering of rocks on land—operate far too slowly to make a difference on a human time-scale. Even if CO2 emissions were somehow to cease today, it would take tens of thousands of years for ocean chemistry to return to its pre-industrial condition.
Acidification has myriad effects. By favoring some marine microbes over others, it is likely to alter the availability of key nutrients like iron and nitrogen. For similar reasons it may let more sunlight penetrate the sea surface. By changing the basic chemistry of seawater, acidification is also expected to reduce the water's ability to absorb and muffle low-frequency sound by up to 40 percent, making some parts of the ocean noisier. Finally, acidification interferes with reproduction in some species and with the ability of others—the so-called calcifiers—to form shells and stony skeletons of calcium carbonate. These last effects are the best documented ones, but whether they will prove the most significant in the long run is unclear.
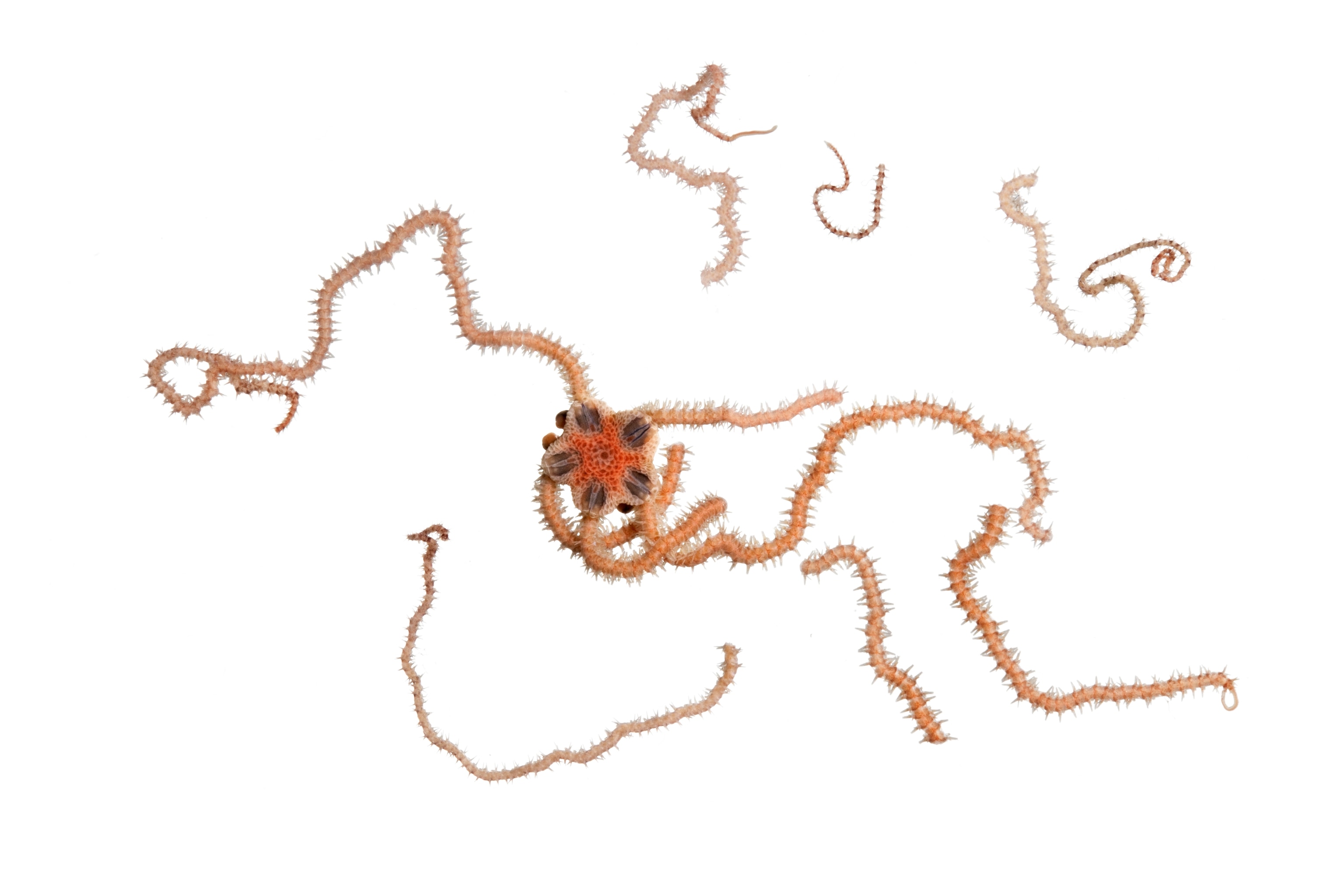
In 2008 a group of more than 150 leading researchers issued a declaration stating that they were "deeply concerned by recent, rapid changes in ocean chemistry," which could within decades "severely affect marine organisms, food webs, biodiversity, and fisheries." Warm-water coral reefs are the prime worry. But because carbon dioxide dissolves more readily in cold water, the impact may actually show up first closer to the Poles. Scientists have already documented significant effects on pteropods—tiny swimming snails that are an important food for fish, whales, and birds in both the Arctic and the Antarctic. Experiments show that pteropod shells grow more slowly in acidified seawater.
Will organisms be able to adapt to the new ocean chemistry? The evidence from Castello Aragonese is not encouraging. The volcanic vents have been pouring CO2 into the water for at least a thousand years, Hall-Spencer told me when I visited. But the area where the pH is 7.8—the level that may be reached oceanwide by the end of the century—is missing nearly a third of the species that live nearby, outside the vent system. Those species have had "generations on generations to adapt to these conditions," Hall-Spencer said, "yet they're not there.
"Because it's so important, we humans put a lot of energy into making sure that the pH of our blood is constant," he went on. "But some of these lower organisms, they don't have the physiology to do that. They've just got to tolerate what's happening outside. And so they get pushed beyond their limits."
Fifty miles off the coast of Australia and half a world away from Castello Aragonese lies the equally tiny One Tree Island. One Tree, which actually has several hundred trees, is shaped like a boomerang, with two arms that stretch out into the Coral Sea. In the crook of the boomerang there's a small research station run by the University of Sydney. As it happened, just as I arrived one spectacular summer afternoon, an enormous loggerhead turtle heaved herself up onto the beach in front of the lab buildings. The island's entire human population—11 people, not including me—gathered around to watch.
One Tree Island is part of the Great Barrier Reef, the world's largest reef complex, which stretches for more than 1,400 miles. The entire island is composed of bits of coral rubble, ranging from marble to basketball size, that began piling up after a peculiarly violent storm about 4,000 years ago. Even today, the island has nothing that could really be called dirt. The trees seem to rise up directly out of the rubble like flagpoles.
When scientists first started visiting the island in the 1960s, they posed questions like, How do reefs grow? Nowadays the questions are more urgent. "Something like 25 percent of all species in the oceans spend at least part of their life in coral reef systems," Ken Caldeira, an expert on ocean acidification at the Carnegie Institution, said one evening before heading out to collect water samples on the reef. "Corals build the architecture of the ecosystem, and it's pretty clear if they go, the whole ecosystem goes."
Coral reefs are already threatened by a wide array of forces. Rising water temperatures are producing more frequent "bleaching" events, when corals turn a stark white and often die. Overfishing removes grazers that keep reefs from being overgrown with algae. Agricultural runoff fertilizes algae, further upsetting reef ecology. In the Caribbean some formerly abundant coral species have been devastated by an infection that leaves behind a white band of dead tissue. Probably owing to all these factors, coral cover in the Caribbean declined by around 80 percent between 1977 and 2001.
Ocean acidification adds yet another threat, one that may be less immediate but ultimately more devastating to hard, reef-building corals. It undermines their basic, ancient structure—the stony skeleton that's secreted by millions upon millions of coral polyps over thousands of years.
Coral polyps are tiny animals that form a thin layer of living tissue on the surface of a reef. They're shaped a bit like flowers, with six or more tentacles that capture food and feed it to a central mouth. (Many corals actually get most of their food from algae that live and photosynthesize inside them; when corals bleach, it's because stress has prompted the polyps to expel those dark symbionts.) Each polyp surrounds itself with a protective, cup-shaped exoskeleton of calcium carbonate that contributes to the collective skeleton of the whole colony.
To make calcium carbonate, corals need two ingredients: calcium ions and carbonate ions. Acids react with carbonate ions, in effect tying them up. So as atmospheric CO2 levels rise, carbonate ions become scarcer in the water, and corals have to expend more energy to collect them. Under lab conditions coral skeleton growth has been shown to decline pretty much linearly as the carbonate concentration drops off.
Slow growth may not matter much in the lab. Out in the ocean, though, reefs are constantly being picked at by other organisms, both large and small. (When I went snorkeling off One Tree Island, I could hear parrotfish chomping away at the reef.) "A reef is like a city," said Ove Hoegh-Guldberg, who used to direct the One Tree Island Research Station and now heads the Global Change Institute at Australia's University of Queensland. "You've got construction firms and you've got demolition firms. By restricting the building materials that go to the construction firms, you tip the balance toward destruction, which is going on all the time, even on a healthy reef. In the end you wind up with a city that destroys itself."
By comparing measurements made in the 1970s with those taken more recently, Caldeira's team found that at one location on the northern tip of the reef, calcification had declined by 40 percent. (The team was at One Tree to repeat this study at the southern tip of the reef.) A different team using a different method has found that the growth of Porites corals, which form massive, boulderlike clumps, declined 14 percent on the Great Barrier Reef between 1990 and 2005.
Ocean acidification seems to affect corals' ability to produce new colonies as well. Corals can, in effect, clone themselves, and an entire colony is likely to be made up of genetically identical polyps. But once a year, in summer, many species of coral also engage in "mass spawning," a kind of synchronized group sex. Each polyp produces a beadlike pink sac that contains both eggs and sperm. On the night of the spawning all the polyps release their sacs into the water. So many sacs are bobbing around that the waves seem to be covered in a veil of mauve.
Selina Ward, a researcher at the University of Queensland, has been studying coral reproduction on Heron Island, about ten miles west of One Tree, for the past 16 years. I met up with her just a few hours before the annual spawning event. She was keeping tabs on a dozen tanks of gravid corals, like an obstetrician making the rounds of a maternity ward. As soon as the corals released their pink sacs, she was planning to scoop them up and subject them to different levels of acidification. Her results so far suggest that lower pH leads to declines in fertilization, in larval development, and also in settlement—the stage at which the coral larvae drop out of the water column, attach themselves to something solid, and start producing new colonies. "And if any of those steps doesn't work, you're not going to get replacement corals coming into your system," Ward said.
The reefs that corals maintain are crucial to an incredible diversity of organisms. Somewhere between one and nine million marine species live on or around coral reefs. These include not just the fancifully colored fish and enormous turtles that people visit reefs to see, but also sea squirts and shrimps, anemones and clams, sea cucumbers and worms—the list goes on and on. The nooks and crevices on a reef provide homes for many species, which in turn provide resources for many others.
Once a reef can no longer grow fast enough to keep up with erosion, this community will crumble. "Coral reefs will lose their ecological functionality," Jack Silverman, a member of Caldeira's team at One Tree, told me. "They won't be able to maintain their framework. And if you don't have a building, where are the tenants going to live?" That moment could come by 2050. Under the business-as-usual emissions scenario, CO2 concentrations in the atmosphere will be roughly double what they were in preindustrial times. Many experiments suggest that coral reefs will then start to disintegrate.
"Under business as usual, by mid-century things are looking rather grim," Caldeira said. He paused for a moment. "I mean, they're looking grim already."
Corals, of course, are just one kind of calcifier. There are thousands of others. Crustaceans like barnacles are calcifiers, and so are echinoderms like sea stars and sea urchins and mollusks like clams and oysters. Coralline algae—minute organisms that produce what looks like a coating of pink or lilac paint—are also calcifiers. Their calcium carbonate secretions help cement coral reefs together, but they're also found elsewhere—on sea grass at Castello Aragonese, for instance. It was their absence from the grass near the volcanic vents that made it look so green.
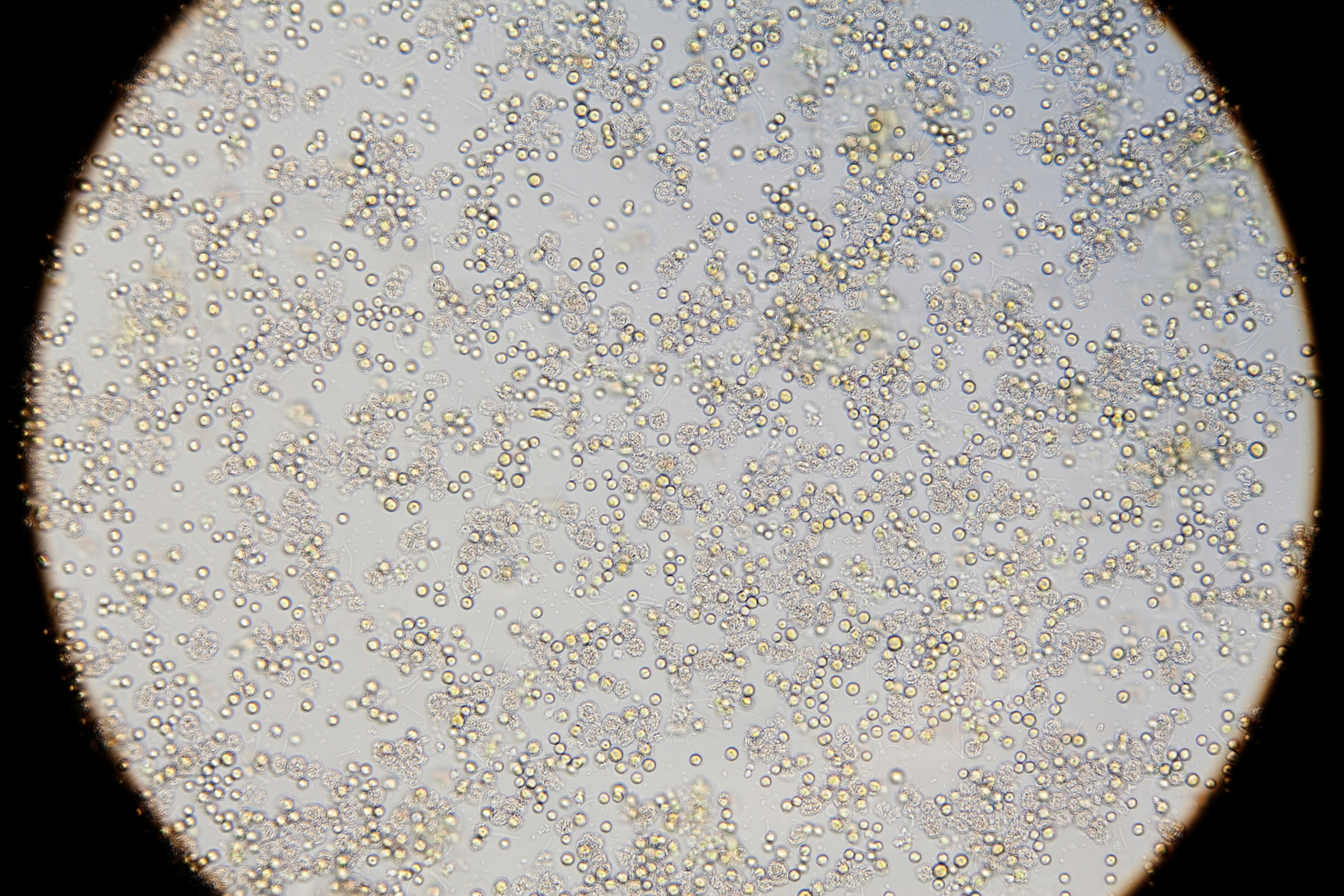
The seas are filled with one-celled calcifying plants called coccolithophores, whose seasonal blooms turn thousands of square miles of ocean a milky hue. Many species of planktonic foraminifera—also one-celled—are calcifiers; their dead shells drift down to the ocean floor in what's been described as a never ending rain. Calcifiers are so plentiful they've changed the Earth's geology. England's White Cliffs of Dover, for example, are the remains of countless ancient calcifiers that piled up during the Cretaceous period.
Acidification makes all calcifiers work harder, though some seem better able to cope. In experiments on 18 species belonging to different taxonomic groups, researchers at the Woods Hole Oceanographic Institution found that while a majority calcified less when CO2was high, some calcified more. One species—blue mussels—showed no change, no matter how acidified the water.
"Organisms make choices," explained Ulf Riebesell, a biological oceanographer at the Leibniz Institute of Marine Sciences in Kiel, Germany. "They sense the change in their environment, and some of them have the ability to compensate. They just have to invest more energy into calcification. They choose, 'OK, I'll invest less in reproduction' or 'I'll invest less in growth.'" What drives such choices, and whether they're viable over the long term, is not known; most studies so far have been performed on creatures living for a brief time in tanks, without other species that might compete with them. "If I invest less in growth or in reproduction," Riebesell went on, "does it mean that somebody else who does not have to make this choice, because they are not calcifying, will win out and take my spot?"
Meanwhile, scientists are just beginning to explore the way that ocean acidification will affect more-complex organisms such as fish and marine mammals. Changes at the bottom of the marine food web—to shell-forming pteropods, say, or coccolithophores—will inevitably affect the animals higher up. But altering oceanic pH is also likely to have a direct impact on their physiology. Researchers in Australia have found, for example, that young clownfish—the real-life versions of Nemo—can't find their way to suitable habitat when CO2 is elevated. Apparently the acidified water impairs their sense of smell.
During the long history of life on Earth, atmospheric carbon dioxide levels have often been higher than they are today. But only very rarely—if ever—have they risen as quickly as right now. For life in the oceans, it's probably the rate of change that matters.
To find a period analogous to the present, you have to go back at least 55 million years, to what's known as the Paleocene-Eocene Thermal Maximum or PETM. During the PETM huge quantities of carbon were released into the atmosphere, from where, no one is quite sure. Temperatures around the world soared by around ten degrees Fahrenheit, and marine chemistry changed dramatically. The ocean depths became so corrosive that in many places shells stopped piling up on the seafloor and simply dissolved. In sediment cores the period shows up as a layer of red clay sandwiched between two white layers of calcium carbonate. Many deepwater species of foraminifera went extinct.
Surprisingly, though, most organisms that live near the sea surface seem to have come through the PETM just fine. Perhaps marine life is more resilient than the results from places like Castello Aragonese and One Tree Island seem to indicate. Or perhaps the PETM, while extreme, was not as extreme as what's happening today.
The sediment record doesn't reveal how fast the PETM carbon release occurred. But modeling studies suggest it took place over thousands of years—slow enough for the chemical effects to spread through the entire ocean to its depths. Today's rate of emissions seems to be roughly ten times as fast, and there's not enough time for the water layers to mix. In the coming century acidification will be concentrated near the surface, where most marine calcifiers and all tropical corals reside. "What we're doing now is quite geologically special," says climate scientist Andy Ridgwell of the University of Bristol, who has modeled the PETM ocean.
Just how special is up to us. It's still possible to avert the most extreme acidification scenarios. But the only way to do this, or at least the only way anyone has come up with so far, is to dramatically reduce CO2 emissions. At the moment, corals and pteropods are lined up against a global economy built on cheap fossil fuels. It's not a fair fight.