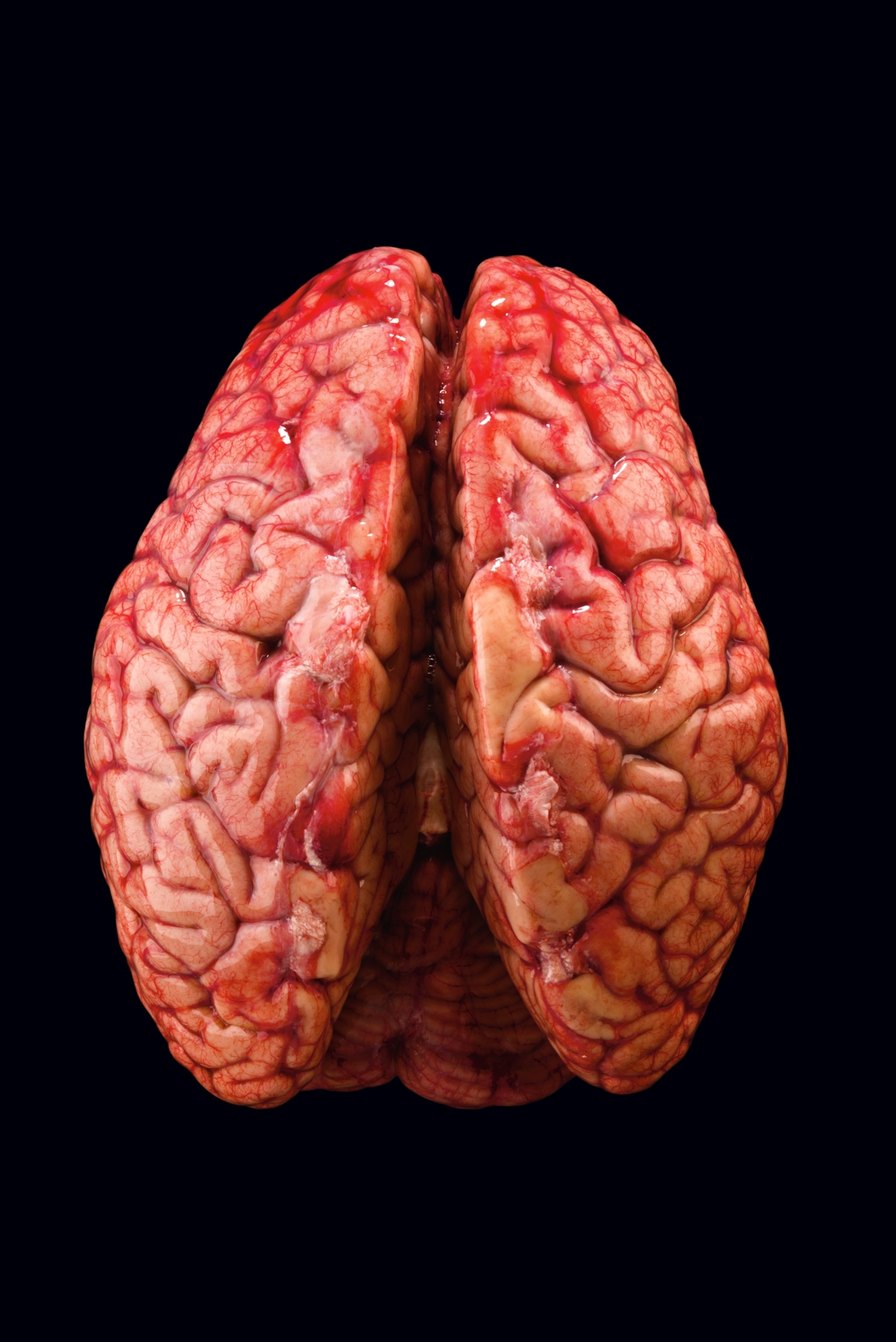
Secrets of the Brain
New technologies are shedding light on biology’s greatest unsolved mystery: how the brain really works.
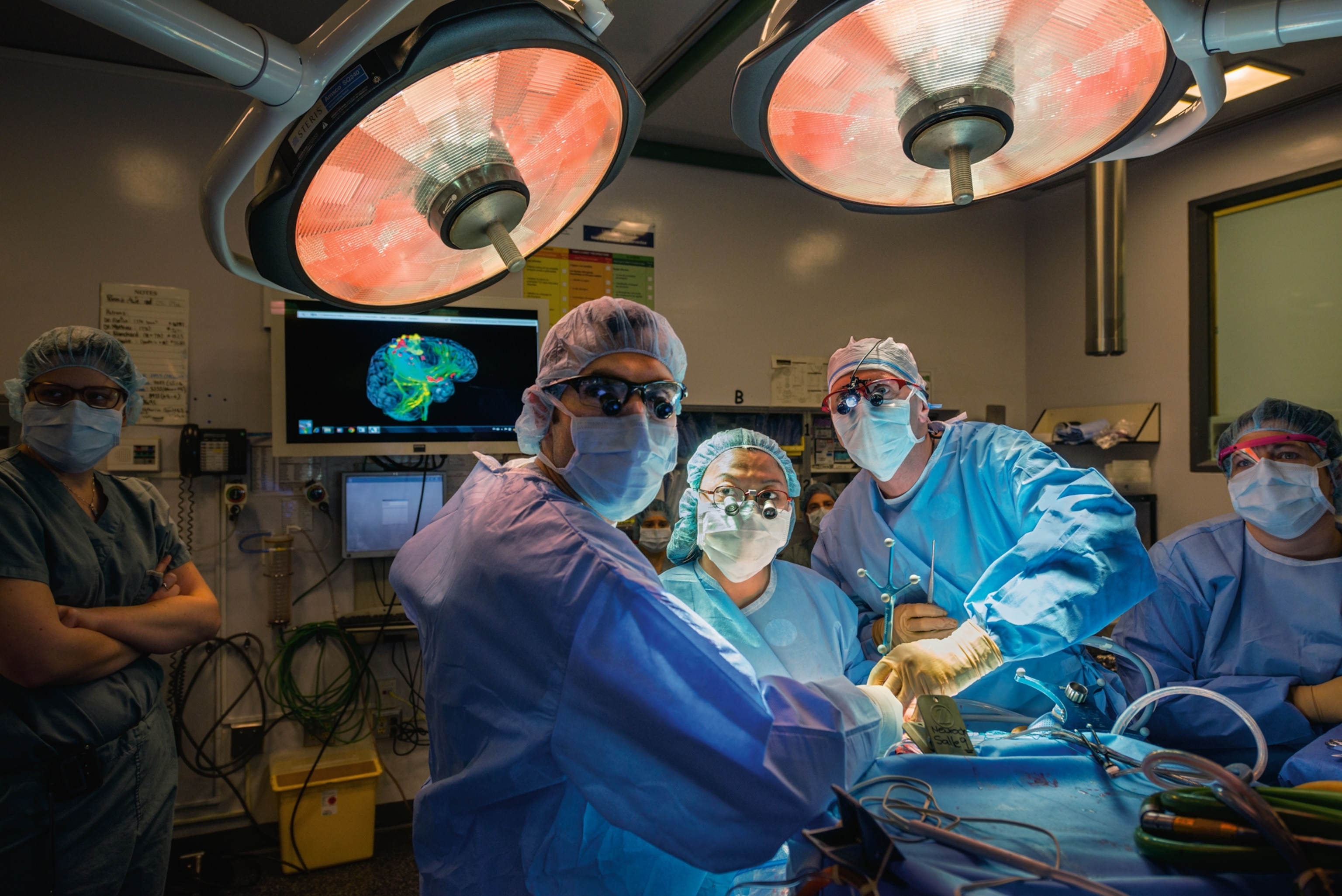
Van Wedeen strokes his half-gray beard and leans toward his computer screen, scrolling through a cascade of files. We’re sitting in a windowless library, surrounded by speckled boxes of old letters, curling issues of scientific journals, and an old slide projector that no one has gotten around to throwing out.
“It’ll take me a moment to locate your brain,” he says.
On a hard drive Wedeen has stored hundreds of brains—exquisitely detailed 3-D images from monkeys, rats, and humans, including me. Wedeen has offered to take me on a journey through my own head.
“We’ll hit all the tourist spots,” he promises, smiling.
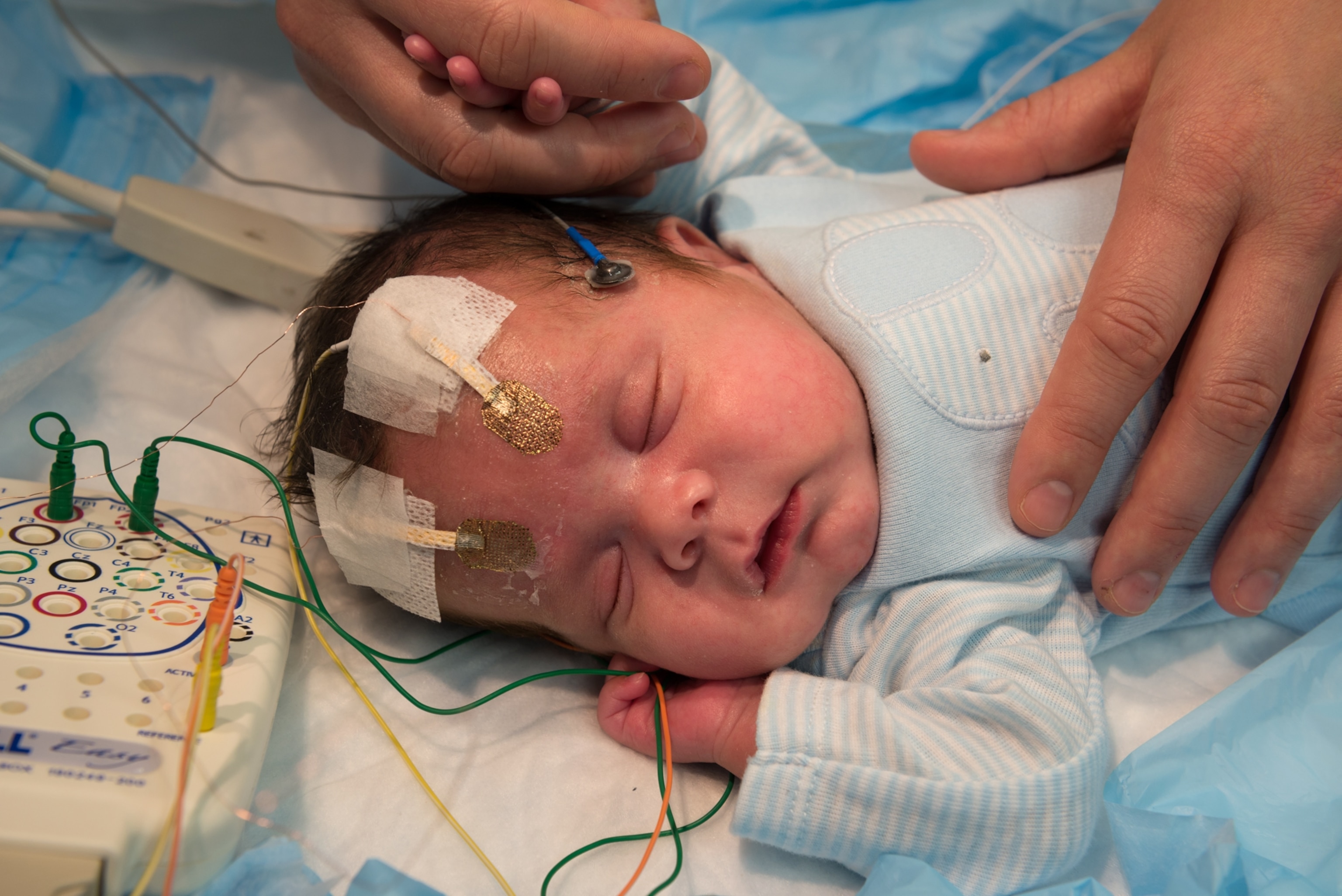
This is my second trip to the Martinos Center for Biomedical Imaging, located in a former ship-rope factory on Boston Harbor. The first time, a few weeks ago, I offered myself as a neuroscientific guinea pig to Wedeen and his colleagues. In a scanning room I lay down on a slab, the back of my head resting in an open plastic box. A radiologist lowered a white plastic helmet over my face. I looked up at him through two eyeholes as he screwed the helmet tight, so that the 96 miniature antennas it contained would be close enough to my brain to pick up the radio waves it was about to emit. As the slab glided into the cylindrical maw of the scanner, I thought of The Man in the Iron Mask.

The magnets that now surrounded me began to rumble and beep. For an hour I lay still, eyes closed, and tried to keep myself calm with my own thoughts. It wasn’t easy. To squeeze as much resolution as possible out of the scanner, Wedeen and his colleagues had designed the device with barely enough room for a person of my build to fit inside. To tamp down the panic, I breathed smoothly and transported myself to places in my memory, at one point recalling how I had once walked my nine-year-old daughter to school through piles of blizzard snow.
As I lay there, I reflected on the fact that all of these thoughts and emotions were the creation of the three-pound loaf of flesh that was under scrutiny: my fear, carried by electrical impulses converging in an almond-shaped chunk of tissue in my brain called the amygdala, and the calming response to it, marshaled in regions of my frontal cortex. My memory of my walk with my daughter was coordinated by a seahorse-shaped fold of neurons called the hippocampus, which reactivated a vast web of links throughout my brain that had first fired when I had clambered over the snowbanks and formed those memories.
I was submitting to this procedure as part of my cross-country reporting to chronicle one of the great scientific revolutions of our times: the stunning advances in understanding the workings of the human brain. Some neuroscientists are zooming in on the fine structure of individual nerve cells, or neurons. Others are charting the biochemistry of the brain, surveying how our billions of neurons produce and employ thousands of different kinds of proteins. Still others, Wedeen among them, are creating in unprecedented detail representations of the brain’s wiring: the network of some 100,000 miles of nerve fibers, called white matter, that connects the various components of the mind, giving rise to everything we think, feel, and perceive. The U.S. government is throwing its weight behind this research through the Brain Research through Advancing Innovative Neurotechnologies (BRAIN) Initiative. In an announcement last spring President Barack Obama said that the large-scale project aimed to speed up the mapping of our neural circuitry, “giving scientists the tools they need to get a dynamic picture of the brain in action.”
As they see the brain in action, neuroscientists can also see its flaws. They are starting to identify differences in the structure of ordinary brains and brains of people with disorders such as schizophrenia, autism, and Alzheimer’s disease. As they map the brain in greater detail, they may learn how to diagnose disorders by their effect on anatomy, and perhaps even understand how those disorders arise.
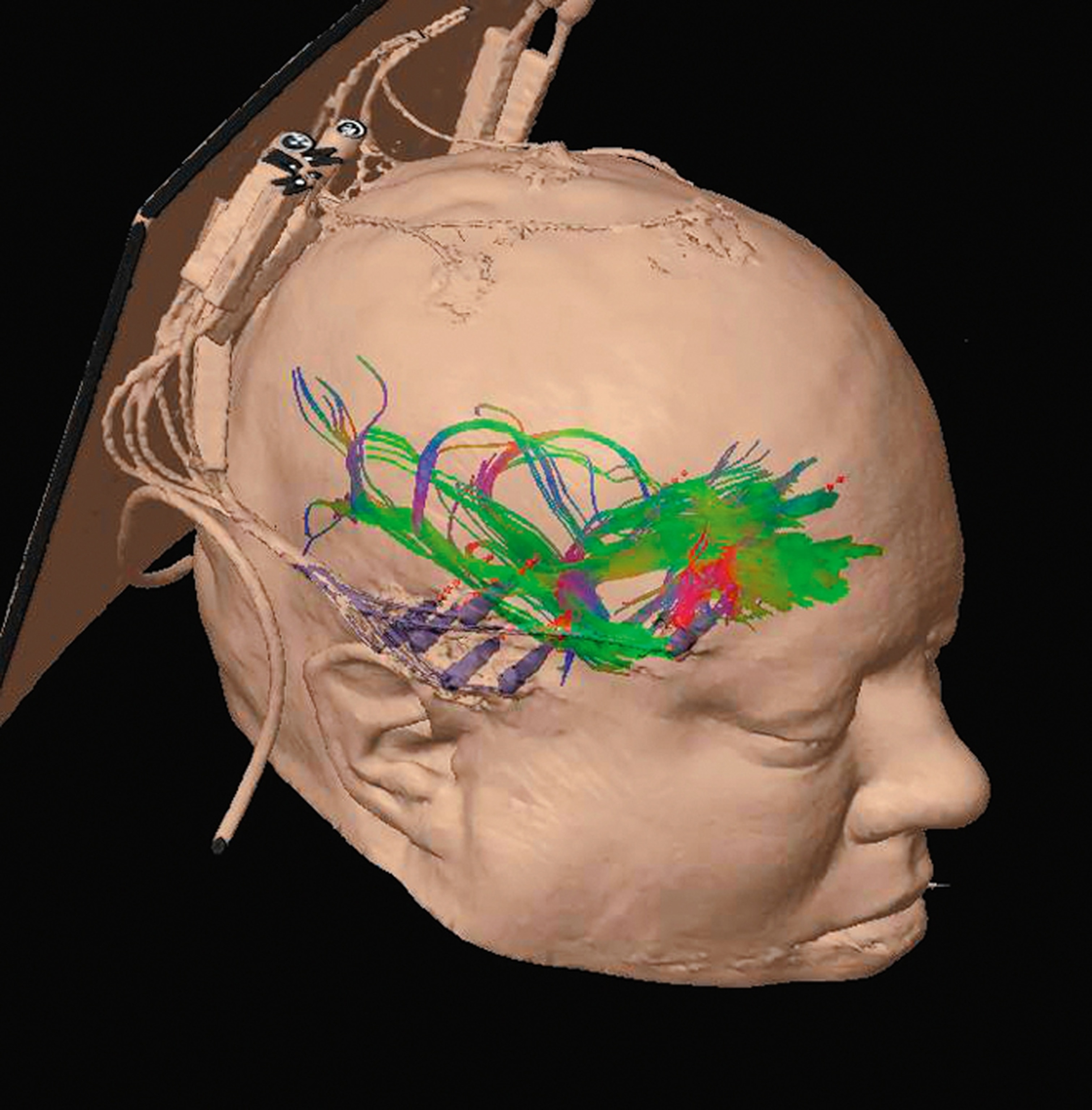
On my return trip to his lab Wedeen finally locates the image from my session in the scanner. My brain appears on his screen. His technique, called diffusion spectrum imaging, translates radio signals given off by the white matter into a high-resolution atlas of that neurological Internet. His scanner maps bundles of nerve fibers that form hundreds of thousands of pathways carrying information from one part of my brain to another. Wedeen paints each path a rainbow of colors, so that my brain appears as an explosion of colorful fur, like a psychedelic Persian cat.
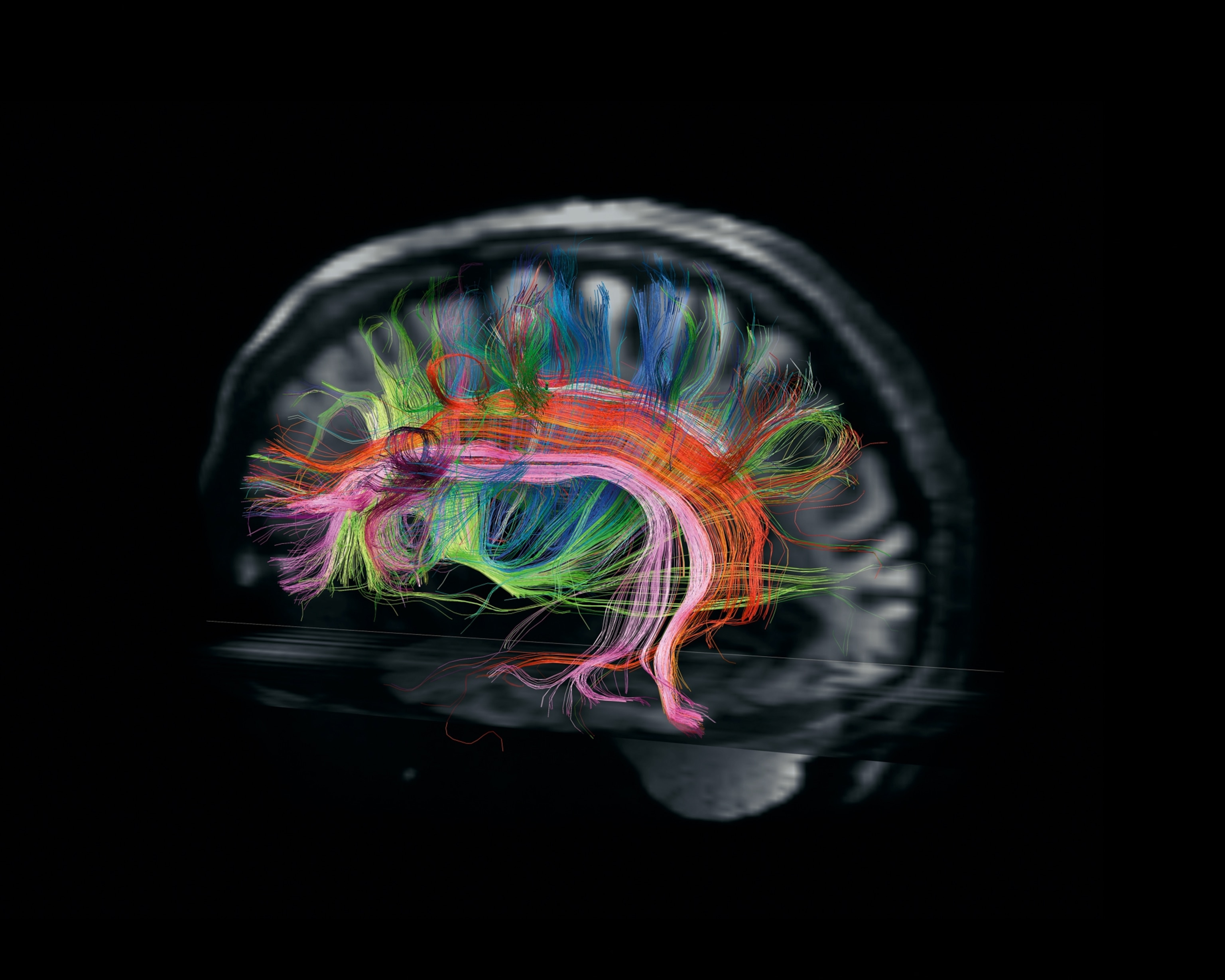

Wedeen focuses in on particular pathways, showing me some of the circuitry important to language and other kinds of thought. Then he pares away most of the pathways in my brain, so that I can more easily see how they’re organized. As he increases the magnification, something astonishing takes shape before me. In spite of the dizzying complexity of the circuits, they all intersect at right angles, like the lines on a sheet of graph paper.
“It’s all grids,” says Wedeen.
When Wedeen first unveiled the grid structure of the brain, in 2012, some scientists were skeptical, wondering if he’d uncovered only part of a much more tangled anatomy. But Wedeen is more convinced than ever that the pattern is meaningful. Wherever he looks—in the brains of humans, monkeys, rats—he finds the grid. He notes that the earliest nervous systems in Cambrian worms were simple grids—just a pair of nerve cords running from head to tail, with runglike links between them. In our own lineage the nerves at the head end exploded into billions but still retained that gridlike structure. It’s possible that our thoughts run like streetcars along these white matter tracks as signals travel from one region of the brain to another.
“There’s zero chance that there are not principles lurking in this,” says Wedeen, peering intently at the image of my brain. “We’re just not yet in a position to see the simplicity.”
Scientists are learning so much about the brain now that it’s easy to forget that for much of history we had no idea at all how it worked or even what it was. In the ancient world physicians believed that the brain was made of phlegm. Aristotle looked on it as a refrigerator, cooling off the fiery heart. From his time through the Renaissance, anatomists declared with great authority that our perceptions, emotions, reasoning, and actions were all the result of “animal spirits”—mysterious, unknowable vapors that swirled through cavities in our head and traveled through our bodies.
The scientific revolution in the 17th century began to change that. The British physician Thomas Willis recognized that the custardlike tissue of the brain was where our mental world existed. To understand how it worked, he dissected brains of sheep, dogs, and expired patients, producing the first accurate maps of the organ.
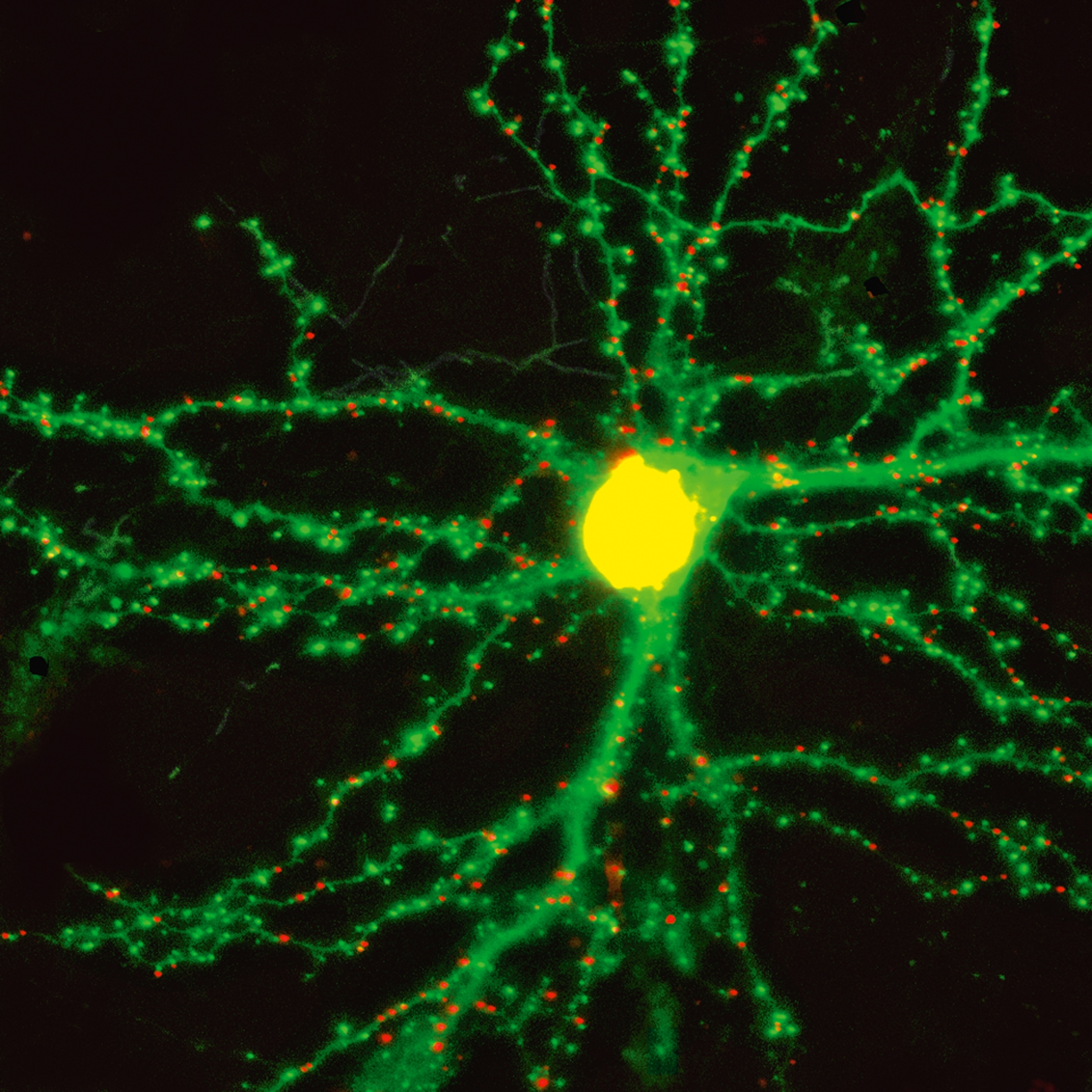
It would take another century for researchers to grasp that the brain is an electric organ. Instead of animal spirits, voltage spikes travel through it and out into the body’s nervous system. Still, even in the 19th century scientists knew little about the paths those spikes followed. The Italian physician Camillo Golgi argued that the brain was a seamless connected web. Building on Golgi’s research, the Spanish scientist Santiago Ramón y Cajal tested new ways of staining individual neurons to trace their tangled branches. Cajal recognized what Golgi did not: that each neuron is a distinct cell, separate from every other one. A neuron sends signals down tendrils known as axons. A tiny gap separates the ends of axons from the receiving ends of neurons, called dendrites. Scientists would later discover that axons dump a cocktail of chemicals into the gap to trigger a signal in the neighboring neuron.
Jeff Lichtman, a neuroscientist, is the current Ramón y Cajal Professor of Arts and Sciences at Harvard, carrying Cajal’s project into the 21st century. Instead of making pen-and-ink drawings of neurons stained by hand, he and his colleagues are creating extremely detailed three-dimensional images of neurons, revealing every bump and stalk branching from them. By burrowing down to the fine structure of individual nerve cells, they may finally get answers to some of the most basic questions about the nature of the brain. Each neuron has on average 10,000 synapses. Is there some order to their connections to other neurons, or are they random? Do they prefer linking to one type of neuron over others?
To produce the images, Lichtman and his colleagues load pieces of preserved mouse brain into a neuroanatomical version of a deli meat slicer, which pares off layers of tissue, each less than a thousandth the thickness of a strand of human hair. The scientists use an electron microscope to take a picture of each cross section, then use a computer to order them into a stack. Slowly a three-dimensional image takes shape—one that the scientists can explore as if they were in a submarine traveling through an underwater kelp forest.
“Everything is revealed,” says Lichtman.
The only problem is the sheer enormity of “everything.” So far the largest volume of a mouse’s brain that Lichtman and his colleagues have managed to re-create is about the size of a grain of salt. Its data alone total a hundred terabytes, the amount of data in about 25,000 high-definition movies.
Once the scientists have gathered this information, the really hard work begins: looking for the rules that organize the brain’s seeming chaos. Recently Lichtman’s postdoctoral researcher Narayanan Kasthuri set out to analyze every detail in a cylinder of mouse brain tissue measuring just a thousand cubic microns—a volume 1/100,000 the size of a grain of salt. He selected a region surrounding a short segment of a single axon, seeking to identify every neuron that passed through it.

That minuscule patch of brain turned out to be like a barrel of seething snakes. Kasthuri found a thousand axons and about 80 dendrites, each making about 600 connections with other neurons inside the cylinder. “It’s a wake-up call to how much more complicated brains are than the way we think about them,” says Lichtman.
Complicated, but not random. Lichtman and Kasthuri discovered that every neuron made nearly all its connections with just one other one, scrupulously avoiding a connection with almost all the other neurons packed tightly around it. “They seem to care who they’re connected to,” Lichtman says.
Lichtman can’t say yet whether this fastidious pattern is a general rule or a feature of just the tiny area of mouse brain he sampled. Even as they scale up the technology, he and his colleagues will need another two years to complete a scan of all 70 million neurons in a mouse. I ask about scanning an entire human brain, which contains a thousand times more neurons than a mouse’s.
“I don’t dwell on that,” he says, with a laugh. “It’s too painful.”
When and if Lichtman completes his three-dimensional portrait of the brain, it will reveal much—but it will still be only an exquisitely detailed sculpture. His imaged neurons are hollow models; real neurons are crammed with living DNA, proteins, and other molecules. Each type of neuron uses a distinct set of genes to build the molecular machinery it needs to do its own job. Light-sensitive neurons in the eyes produce photon-catching proteins, for example, and neurons in a region called the substantia nigra produce dopamine, crucial to our sense of reward. The geography of proteins and other chemicals is essential to understanding how the brain works—and how it goes awry. In Parkinson’s disease the substantia nigra neurons produce less dopamine than normal, for reasons that aren’t yet clear. Alzheimer’s disease scatters tangles of protein through the brain, although scientists have yet to firmly settle on how those tangles give rise to the devastating dementia the disease causes.
A map of the brain’s molecular machinery called the Allen Brain Atlas has been generated at the Allen Institute for Brain Science in Seattle, founded ten years ago with funds from Microsoft co-founder Paul Allen. Using the brains of recently deceased people, donated by their families, researchers there use a high-resolution magnetic resonance imaging (MRI) scan of each brain as a three-dimensional road map, then slice it into microscopically thin sections that are mounted on glass slides. They then douse the sections with chemicals that reveal the presence of active genes harbored in the neurons.
So far the researchers have mapped the brains of six people, charting the activity of 20,000 protein-coding genes at 700 sites within each brain. It’s a colossal amount of data, and they’ve only begun to make sense of it. The scientists estimate that 84 percent of all the genes in our DNA become active somewhere in the adult brain. (A simpler organ like the heart or pancreas requires far fewer genes to work.) In each of the 700 sites the scientists studied, the neurons switch on a distinct collection of genes. In a preliminary survey of two regions of the brain, the scientists compared a thousand genes that were already known to be important for neuron function. From one person to the next, the areas of the brain where each of those genes was active were practically identical. It looks as if the brain has a finely grained genetic landscape, with special combinations of genes carrying out tasks in different locations. The secret to many diseases of the brain may be hiding in that landscape, as certain genes shut down or switch on abnormally.
All the information from the Allen Brain Atlas is posted online, where other scientists can navigate through the data with custom-made software. Already they’re making new discoveries. A team of Brazilian scientists, for instance, has used it to study a devastating brain disorder called Fahr’s disease, which calcifies regions deep inside the brain, leading to dementia. Some cases of Fahr’s disease had already been linked to a mutation in the gene SLC20A2. In the atlas the scientists found thatSLC20A2 is most active in precisely the regions that are targeted by the disease. They also found a network of other genes that is most active in the same areas, and now they’re trying to find out whether they’re involved in Fahr’s disease as well.
Of all the new ways of visualizing the brain, perhaps the most remarkable is one invented by Stanford neuroscientist and psychiatrist Karl Deisseroth and his colleagues. To see the brain, they begin by making it disappear.
On my visit to Deisseroth’s lab, undergraduate Jenelle Wallace led me to a bench where half a dozen beakers rested in a plastic-foam base. She pulled one out and pointed to a grape-size mouse brain resting at the bottom. I didn’t look at the brain so much as through it. It was nearly as transparent as a glass marble.
Needless to say, a normal human or mouse brain is decidedly opaque, its cells swathed in fat and other compounds that block light. That’s why Cajal had to dye neurons in order to see them and why Lichtman’s group and the Allen Institute scientists slice the brain into thin sections to gain access to its inner depths. The advantage of a transparent brain is that it allows us to peer into its workings while the organ is still intact. Along with postdoctoral researcher Kwanghun Chung, Deisseroth came up with a recipe to replace the light-scattering compounds in the brain with transparent molecules. After making a mouse brain transparent in this way, they can then douse the brain with glowing chemical labels that latch on to only certain proteins or trace a specific pathway connecting neurons in distant regions of the brain. The scientists can then wash out one set of chemicals and add another that reveals the location and structure of a different type of neuron—in effect untangling the Gordian knot of neural circuits one by one. “You don’t have to take it apart to show the wiring,” says Deisseroth.
It’s not easy to dazzle neuroscientists, but Deisseroth’s method, dubbed CLARITY, has left his colleagues awestruck. “It’s pretty badass,” says Christof Koch, the chief scientific officer at the Allen Institute. Wedeen has called the research “spectacular ... unlike anything else in the field.”
Because of our shared evolutionary heritage, a clarified mouse brain can reveal a great deal about human brain function. But Deisseroth’s ultimate goal is to perform the same transformation with a human brain—a far more difficult task, not least because a human brain is 3,000 times as large as that of a mouse.
A CLARITY picture showing the location of just one type of protein in just one human brain would create a monstrous heap of data—about two petabytes, or the equivalent of several hundred thousand high-def movies. Deisseroth anticipates that CLARITY may someday help the sort of people he treats in his psychiatric practice, by revealing hidden features of disorders like autism and depression. But for now he’s keeping those hopes in check.
“We have so far to go before we can affect treatments that I tell people, Don’t even think about that yet,” he says. “It’s just a voyage of discovery for now.”
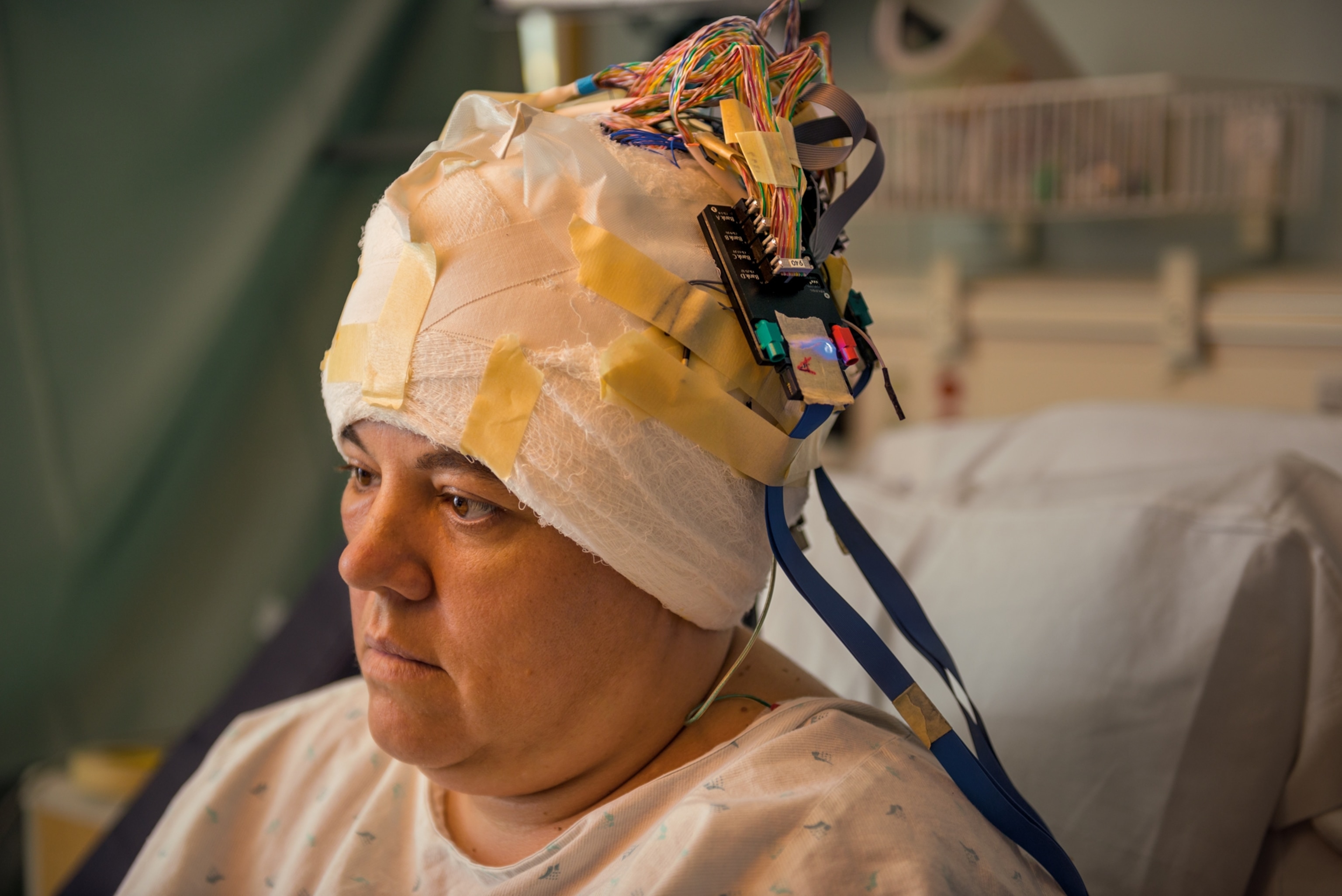
As revealing as a transparent brain may prove to be, it will still be dead. Scientists need different tools to explore the terrain of living brains. The scanners Wedeen uses to trace white matter patterns can, with different programming, record the brain in action. Functional magnetic resonance imaging (fMRI) pinpoints regions of the brain recruited during a mental task. Over the past couple of decades fMRI has helped reveal networks involved in all manner of thought processes, from recognizing faces to enjoying a cup of coffee to remembering a traumatic event.
It’s easy to be dazzled by fMRI images, which festoon the brain with rainbow blobs. But it’s important to bear in mind that those images are actually quite coarse. The most powerful scanners can record activity only down to the scale of a cubic millimeter—a sesame seed’s worth of tissue. Within that space, hundreds of thousands of neurons are firing in synchronized patterns, trading signals. How those signals give rise to the larger patterns revealed by fMRI remains mysterious.
“There are ridiculously simple questions about the cortex that we can’t answer at all,” says Clay Reid, a former colleague of Jeff Lichtman’s at Harvard who moved to the Allen Institute in 2012.
Reid has come to Seattle hoping to answer some of those questions with a grand series of experiments he and his colleagues call MindScope. Their goal is to understand how a large number of neurons carry out a complex task.
The function Reid and his colleagues have chosen to decipher is vision. Scientists have been investigating how we see for decades, but they’ve been able to study it only piecemeal. A neuroscientist might place an electrode in the region of a mouse’s brain involved in visual perception and then note whether nearby neurons fire when the animal sees a particular image.
This approach has allowed scientists to map regions of the visual brain that specialize in different tasks, such as detecting the edges of an object or perceiving brightness. But scientists haven’t been able to see all those regions work together at once—to learn how the million or so neurons in the visual regions of a mouse’s brain instantly put information together into the image of a cat.
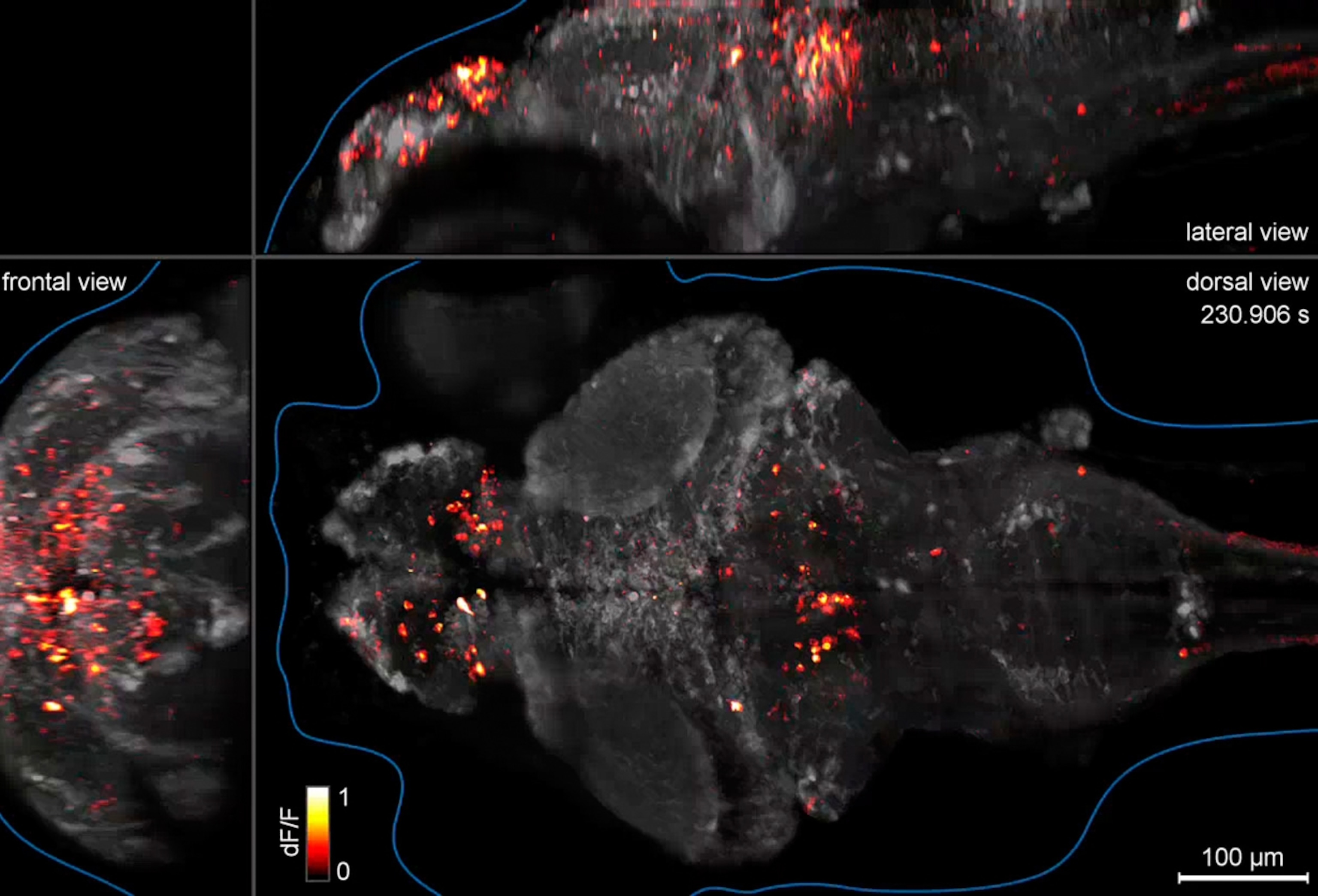
Reid and his colleagues are setting out to solve that problem by engineering mice so that their visual neurons will release flashes of light when they fire. The flashes record the neural activity when a mouse sees a specific object, be it a cat, a snake, or an appealing piece of cheese. The scientists can then compile the data to create massive mathematical models of vision. If the models are accurate, the researchers will be able to literally read the mind of a mouse.
“Our goal is to reconstruct what the mouse sees,” says Reid. “And I think we can do it.”
Reid’s research on mouse vision is another step toward neuroscience’s ultimate goal: a comprehensive view of how this vastly complicated organ really works—what the scientists I talked to call a theory of the brain. Such a grand vision is still a long way off, and for the most part, the search for it has yet to change the way doctors treat patients. But there is one line of research—brain-machine interfaces—where the mapping of the mind has started to change people’s lives.
When she was 43 years old, Cathy Hutchinson suffered a massive stroke, leaving her unable to move or speak. Lying in her bed in Massachusetts General Hospital, she gradually figured out that her doctors didn’t know if she was brain-dead or still aware. Her sister asked Hutchinson if she could understand her. She managed to answer by moving her eyes up on command.
“It gave me such a relief,” Hutchinson tells me 17 years later, “because everybody talked about me as if I was dying.”
It is a chilly winter day at her home in eastern Massachusetts, and she’s sitting in a wheelchair in the middle of the living room, dressed in a dark green jogging suit and sneakers. Still almost completely paralyzed and unable to speak, she communicates by looking at letters arrayed on a computer monitor bolted to her wheelchair, a camera tracking the movement of a tiny metal disk attached to the center of her eyeglasses.
Near the top of the brain is a region called the motor cortex, where we generate commands to move our muscles. For more than a century we’ve known that each part of the cortex corresponds to a particular area of the body. When people like Hutchinson become paralyzed, the motor cortex often remains intact, but it can’t communicate with the rest of the body, because its connections have been destroyed. John Donoghue, a neuroscientist at Brown University, wanted to find a way to help people with paralysis by tapping into the signals from their motor cortex. Perhaps they could eventually learn to type on a computer or operate a machine merely with their thoughts. Donoghue spent years developing an implant and testing the device on monkeys. Once he and his colleagues knew it was safe, they were ready to start working with human patients.
One of them was Hutchinson. In 2005 surgeons at Rhode Island Hospital drilled a hole the size of a poker chip in her skull and inserted the sensor for Donoghue’s device. About the size of a ladybug, the sensor contained a hundred miniature needles, which, pressing into Hutchinson’s motor cortex, recorded the signals from nearby neurons. A set of wires anchored to this device passed through the hole in her skull and led to a metal connector sitting on her scalp.
After her surgery had healed, the Brown University researchers plugged Hutchinson’s implant into a cable that relayed signal patterns from her brain to a cart of computers they wheeled into her room. As a first step, the scientists trained the computers to recognize signals in her motor cortex and use them to move a computer cursor around a screen. This was achieved the first time she tried because they had learned how to translate patterns of brain activity into movements. Two years later they coupled a robotic arm to the computers, refining a program that could interpret Hutchinson’s brain signals to move the arm forward and back, to raise it up and down, and to open its robotic fingers and squeeze them shut.
After just a few sessions Hutchinson, the computer, and the robotic arm had become a team. “It felt natural,” she tells me. So natural that one day she reached out for a cinnamon latte, grabbed it, and brought it to her lips to drink.
“Cathy’s smile when she put down that drink—that’s everything,” Donoghue says.
Today Donoghue and other scientists are building on that success, hoping to create human-machine interfaces that will be powerful, safe, and easy. At Duke University Miguel Nicolelis has been experimenting with exoskeletons that strap on to the body. Signals from the brain control each limb. Already he has gotten monkeys to control full-body exoskeletons. If all goes well, a paraplegic wearing a simpler version of the device will deliver the opening kick at the 2014 World Cup in Nicolelis’s native Brazil.
“Eventually brain implants will become as common as heart implants,” says Nicolelis. “I have no doubt about that.”
When it comes to the brain, predicting the future is a tricky game. Advances in the past have inspired giddy expectations that in many cases have not been met. “We can’t tell a schizophrenic brain from an autistic brain from a normal brain,” says Christof Koch. But the research that’s going on now, he believes, is moving neuroscience to a remarkable new stage. “I think we can begin to put the pieces together.”