
How devastating pandemics change us
Deadly outbreaks have plagued societies for centuries. But they can lead to medical breakthroughs—if we learn the right lessons from them.
On a Sunday early in March, as the COVID-19 outbreak was racing across the planet, the U.S. Coast Guard cutter Pike pitched and rolled en route to the cruise ship Grand Princess, waiting 14 miles off the coast of California. The cutter was delivering a medical disaster team to separate the sick from the seemingly healthy among the 3,500 people aboard and prepare to bring them ashore. On the Pike, Michael Callahan, an infectious disease specialist with decades of experience in ‘hot zones’ everywhere, waited with his team, he says, ‘unheroically’ vomiting.


Shortly before sunset, the Pike approached a tender lowered from the Grand Princess. Callahan, age 57, and his team, still seasick, were now also half-deafened and blinded in full biocontainment gear. One by one, they made the leap first to the tender, and then, as the boat banged against the hull of the 188-foot-tall cruise ship, they leaped again onto a ladder and climbed up the hull to begin their work.
The entire world at that moment was also taking a leap into the unknown. Or rather, into the forgotten. Epidemics always have afflicted humans, and pandemics since we first sprawled across the globe. They have taught us important lessons—if only we could manage to remember them in our exhaustion and relief after danger has passed us by. New pandemics such as COVID-19 have a way of reminding us how easy it is for us to infect one another, especially those we love. How fear of contagion forces us apart. How devastating isolation can be, and yet how the sick often must die miserably and alone. Above all, the new pandemic has reminded us how much we’ve always depended on a small, brave band of people like Callahan—we’ll come back to him—who risk their lives fighting diseases.
Steps along the way
Jump ahead to 1721, Boston | 1800s, London | Late 1800s, Europe | Present day

These people have often been too flawed, too human, to fit the traditional mold of hero. In past pandemics, they have tended to be individuals willing to disregard conventional thinking, to learn from small, seemingly insignificant clues, or to listen to unexpected voices. And they have been willing to recognize that what happened over there, in some dismal neighborhood or some forgotten corner of the world, could easily happen here too. To understand these people who help end pandemics, the best place to start is with one of the worst diseases in human history.
1721, Boston: From inoculation to vaccination
At a lecture in Boston early in 1721, Cotton Mather, the hellfire Puritan minister, announced the coming of “the destroying angel,” a terrifying disease bearing down on the city. England was already under siege.
The New World had felt its dreadful effects before, passing through in unpredictable waves over 200 years, inflicting panic and misery on the colonists and wiping out entire Native American societies. But it had been 19 years since Boston’s last epidemic, time enough to raise a fresh generation of victims.

When the first light-red spots appeared, you could hope it might be just measles. But then the spots turned into bumps, filled with fluid, and rose up like volcanic islands on the skin. Hundreds of them could clot the eyes, the air passages, the entire body, to make even breathing an agony. The pustules gave off the stench of rotting flesh. Survivors were often blinded, crippled, or badly disfigured. (The doctor tending one sick British woman was instructed, “Preserve her beauty, or take her life.”) That April, smallpox slipped quietly into Boston Harbor.
At first, people ignored the outbreak, much as has happened in our own time. But starting in 1721, smallpox taught the Western world a powerful new lesson: Humans can prevent pandemic diseases. We can hem them in and, if we have the will, sometimes even eradicate them. Three unlikely heroes took up the fight that year in Boston. They included the African-born slave Onesimus—a biblical name Mather had put on him—and a physician and surgical innovator named Zabdiel Boylston. But the unlikeliest of them was Mather himself, a troubled character, vain, emotionally unstable, and still widely disliked as a dark force behind the Salem witch trials 29 years earlier.
Now, though, it was as if Mather had been preparing all his life for this moment, and for redemption. He had been a keen student of science and medicine from childhood, and no doubt it also became personal: Two wives and 13 of his 15 children would die before him, many from infectious disease. So he read British science journals and studied Native American medicines. And he paid attention when his “servant” Onesimus, “a pretty Intelligent Fellow,” told him about a method of preventing smallpox in Africa and showed him the resulting scars. Details of this method were also circulating in England, based on reports from Turkey.

As the outbreak was beginning to spread, Mather alerted Boston’s physicians to “a Wonderful Practice lately used in several Parts of the World” to stop it. The technique was to take a patient with smallpox and puncture one of the ripe pustules to draw off pus, or “variolous matter.” A portion of this material then went into an incision in the skin of someone who was still perfectly healthy. The promise of “variolation,” or inoculation, was that it would produce immunity, after what would probably be only a mild case of one of the deadliest diseases on Earth.
Mather found corroborating accounts and scars from the procedure among “a Considerable Number” of other African-born Bostonians. Boston’s medical community recoiled. But Zabdiel Boylston knew the terror of smallpox from having nearly died of it 19 years earlier, and he worried that his medical practice put his eight children “daily in danger.” On June 26, having considered the evidence, he performed his first variolations, on his six-year-old son and two family slaves. The result was “a kind and favourable Small-Pox,” and he began to inoculate patients seeking protection from the full-blown disease.
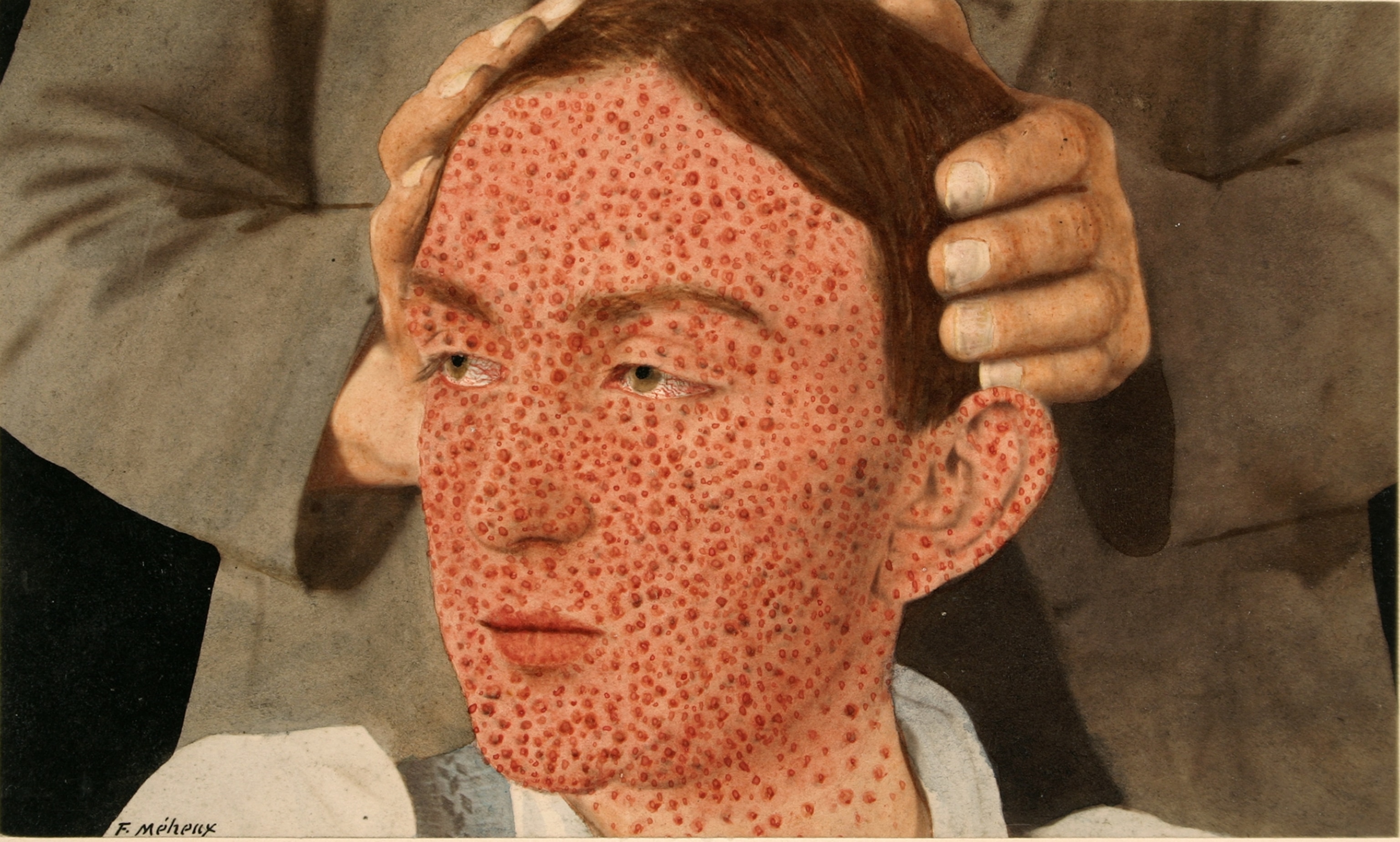
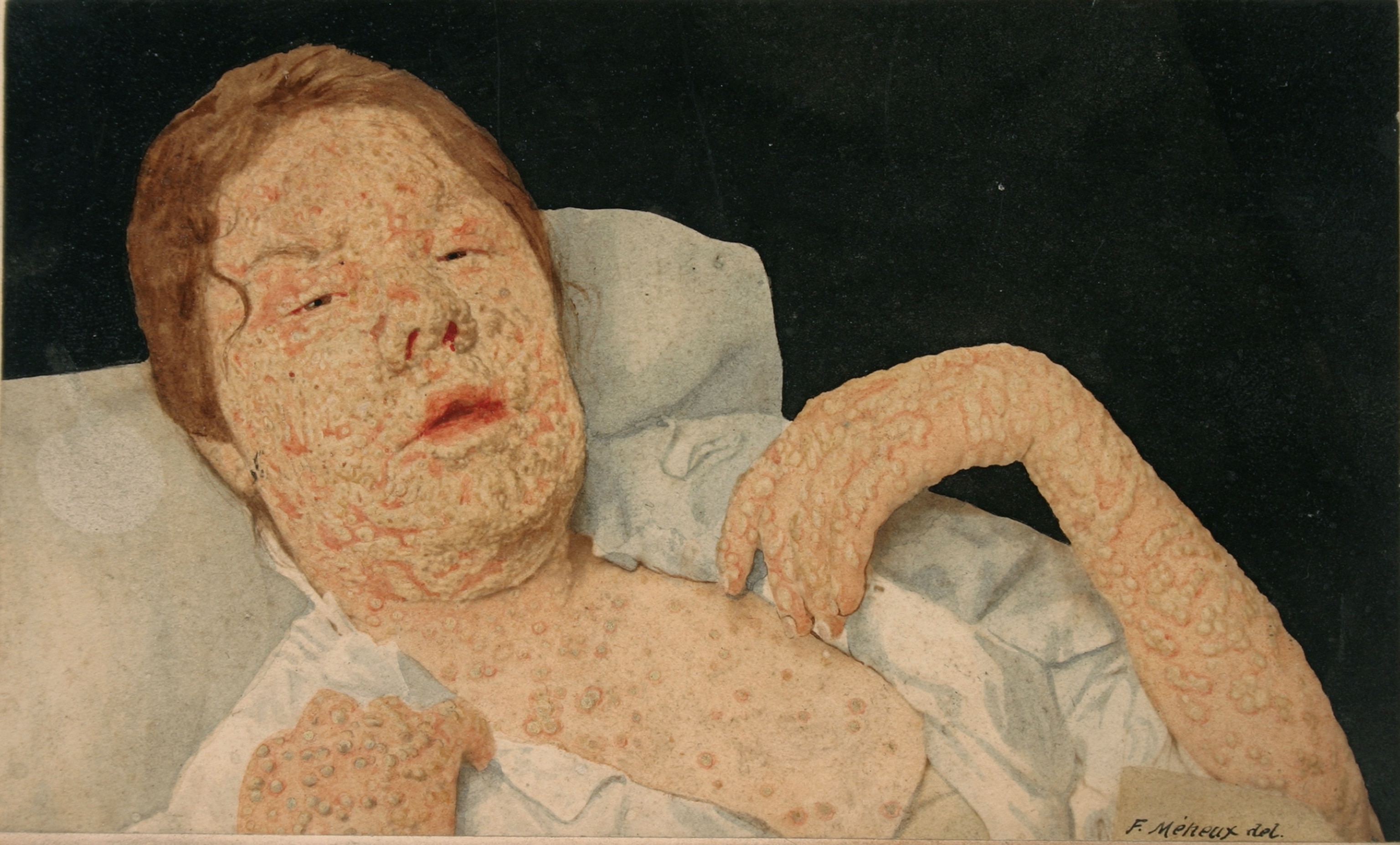
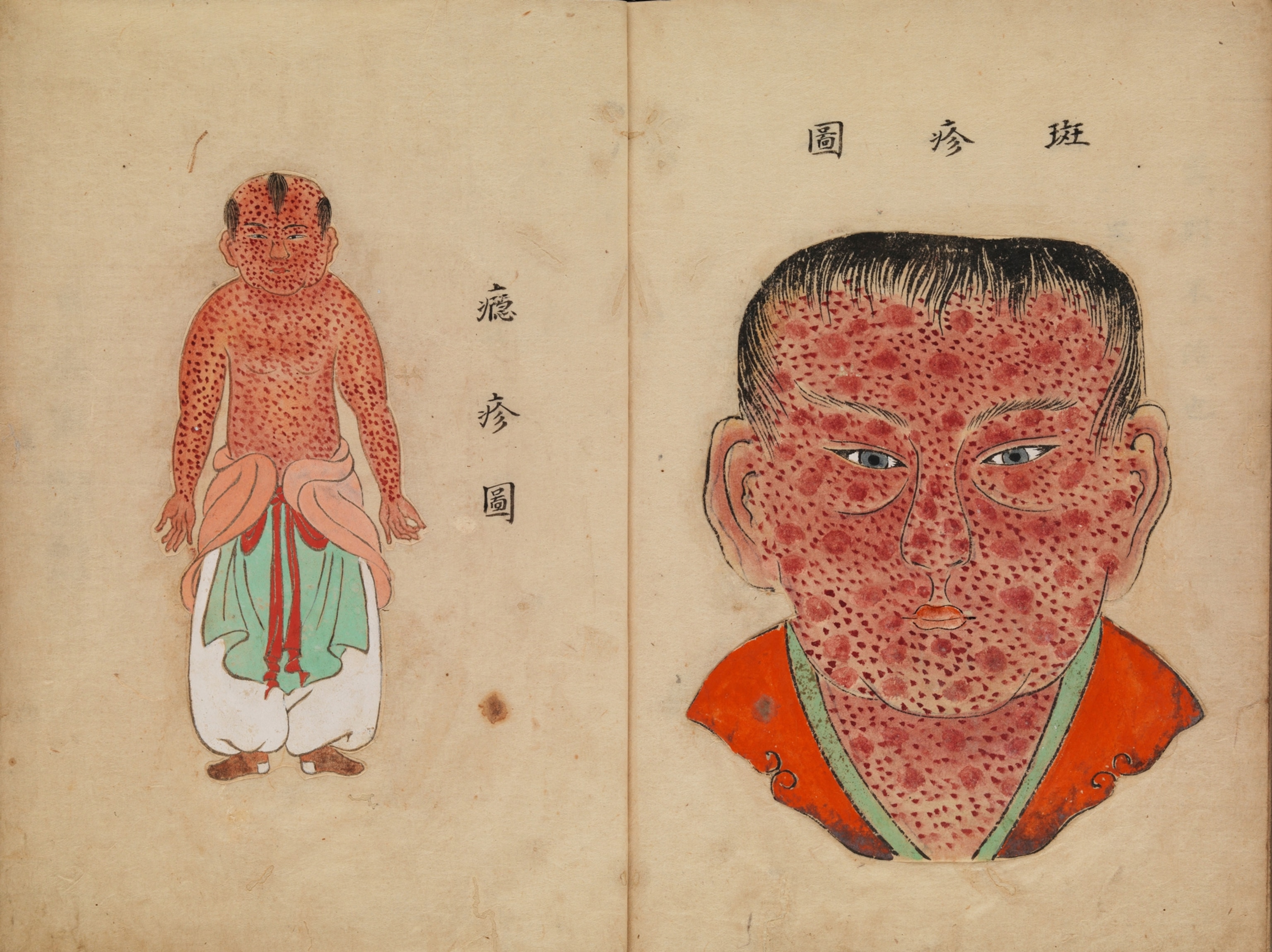
Some city residents considered the treatment as terrifying at first as the disease itself. They worried that variolated patients who hadn’t fully recovered were still contagious. Doctors objected that the practice flew in the face of medical orthodoxy, which had held for 2,000 years that disease resulted from an imbalance in four bodily “humors,” often brought on by foul odors and ill-defined “miasmas,” or bad air.
Tradesmen and laborers couldn’t afford to protect their families because of the high cost of inoculation and the attendant medical care before and after. Terror and class resentment, together with the lingering shadow of the witch trials, helped make Mather a target. One night a firebomb came flying through a bedroom window of his house. By chance, the fuse fell off and the bomb landed with a harmless thud. Tied to it was a note: “COTTON MATHER, You Dog, Dam you: I’ll inoculate you with this, with a Pox to you.”
By the time the epidemic finally ended, almost 6,000 residents, more than half of Boston, had come down with smallpox and 844, about 15 percent, had died. On the other hand, just 2 percent of those who had undergone variolation died. Improvements soon drove that to less than half a percent, and variolation became standard practice. When another smallpox epidemic hit Boston in 1792, the response had completely reversed: About 9,200 local residents were inoculated, and only 232 of them suffered natural smallpox.
None of the three men who introduced variolation to North America won much honor by it. Onesimus disappeared from the record after purchasing his freedom, and the African contribution was swept out of sight. Zabdiel Boylston was also mostly forgotten. Streets, buildings, and a nearby town named Boylston actually honor his grandnephew, a wealthy merchant. Cotton Mather, finally, did not win redemption in Boston hearts. But he continued to think on medical matters, eventually writing about the true cause of all epidemics: In the proper conditions, tiny organisms, which were then just beginning to be seen by microscope, “soon multiply prodigiously; and may have a greater Share in producing many of our Diseases than is commonly imagined.” But his otherwise eccentric manuscript went unpublished. It would take scientists another 150 years to recognize the critical role of microbes as the agents of infectious disease.
Starting in 1721, smallpox taught the Western world a powerful new lesson: Humans can prevent pandemic diseases. We can hem them in and, if we have the will, sometimes even eradicate them.
The push for variolation in North America and Europe produced one other unexpected effect. In 1757, in a small town in the south of England, a “fine ruddy boy” of eight underwent variolation. It was a miserable experience, because doctors bound by tradition demanded a preparatory regimen of bleeding and purging. When the boy became a country doctor himself, “a vague opinion prevailed” among the dairies, he wrote, that a livestock disease called cowpox might be “a preventive of the Small Pox.” The possibility of a better way made a powerful impression on Edward Jenner. But for decades, no one put this rumored treatment into practice, until finally Jenner himself saw his chance.
On May 14, 1796, he performed what looked like variolation on another eight-year-old boy, named James Phipps, but using material from a young woman infected with cowpox. It was the beginning of modern vaccination, a term coined from the Latin vacca, for cow. The first anti-vaxxers immediately rose up in outraged protest. They feared, among other things, that people could develop cow-like tendencies, catch animal diseases, or even sprout horns. But vaccination proved so much safer and more effective than variolation that it soon spread across the Earth.




(See maps that show how smallpox invaded the Americas.)
Smallpox continued to kill people, an estimated 300 million in the 20th century alone. In May 1980 the World Health Organization declared smallpox eradicated thanks to a determined global vaccination campaign. By then, Jenner’s vaccine had become the model for a host of others. They drove so many infectious diseases from our lives that for a brief, happy interlude, it seemed possible that such a thing as a pandemic could never happen again.
1800s, London: Public water and sewers
People were willing to accept the next great lesson in ending pandemics only because of a disease that was among the most frightening they had ever experienced. When this scourge broke out in 1817 in the city of Jessore, now part of Bangladesh, its new virulence shocked people even there, though they knew its terrors from past outbreaks.
“So unforeseen and appalling was the attack,” wrote a district official, that astonished residents “fled in crowds to the country as the only means of escaping impending death.” In just a few weeks, 10,000 people died in this one district.

A booming commercial and colonial trade carried the new outbreak overland and across oceans and made it pandemic. Newspaper readers could follow reports from the front lines as the dreaded disease crept toward them. It wasn’t just that it killed half its victims and did so with appalling speed. A special horror attended the way they died, with a person who was in the prime of life one moment seeming, in the next, to liquefy and flow out in uncontrollable vomiting and diarrhea. Intense thirst followed. Spasms and cramps wrenched the muscles. Breathing became a desperate, gasping “air hunger.” Victims died with their minds seemingly intact, staring, aghast, the watery liquid still being wrung from their guts.
When people debated the cause of this new menace, miasmas and foul odors were the usual suspects. Almost all the early sanitary reformers focused obsessively on smells, partly because they were everywhere—the acrid odors of factories, the pigsties adjacent to homes, the tonnage of droppings from horses and livestock, the tanneries, the shallow graves of the dead, and of course human excrement everywhere. For the sanitary movement, “putrid exhalations” were the cause of disease.
In the 19th century, as people left the farm and lined up for factory jobs in the city, humanity still badly needed lessons in how to live together without dying. Practices that had seemed harmless on the farm, like the lack of organized sewage disposal, proved fatal in cities. Families huddling one atop another in squalid slums circulated and recirculated typhoid fever, dysentery, tuberculosis, cholera, and other infectious diseases.

The great teacher of sanitary reform was a British civil servant named Edwin Chadwick, a Dickensian bureaucrat, tall, round-faced, hair smeared in hanks across his balding scalp, peering out from heavy-lidded eyes in judgment, if not disdain. He was “a really outstanding specimen of bore,” according to one biographer, “in an age when the species flourished.” But he also made a reputation for mastering the facts of any problem he studied and for bringing prodigious energy to the solution.
In 1842 Chadwick wrote an unlikely best seller, published by the British government, now known as The Sanitary Report. Based on accounts from around Britain, it described in unblinking detail an urban working-class world that must have seemed as foreign as Jessore to most educated readers of the era. Chadwick led them into cellars three feet deep in human waste from overflowing cesspools, and houses where “every article of food and drink must be covered” to avoid “the strong taste of the dunghill” carried in by houseflies. He described a town where “the filth” of a jail holding 65 prisoners “is floated down the public streets every second or third day,” joined by blood from the local slaughterhouse.

Chadwick was a believer in “filth theory” and the deadly power of stenches. Luckily, his detailed recommendations also happened to work against the real causes of disease.
(Read about how a Scottish doctor spread the sanitary gospel of filth theory in the mid-1700s.)
The visceral horror of The Sanitary Report roused reluctant politicians to the need to do something. In 1848 the British government established one of the world’s first national public health authorities, with Chadwick in charge. The following year a cholera outbreak unexpectedly cracked the whip on behalf of sanitary reform. Chadwick soon launched a nationwide campaign pushing cities and towns to build centralized public systems to supply clean water to homes, together with properly designed sewerage to carry away wastes. It was a massively expensive undertaking but produced dramatic improvements in health and life span. Other nations followed, and for the first time cities began to become truly livable.

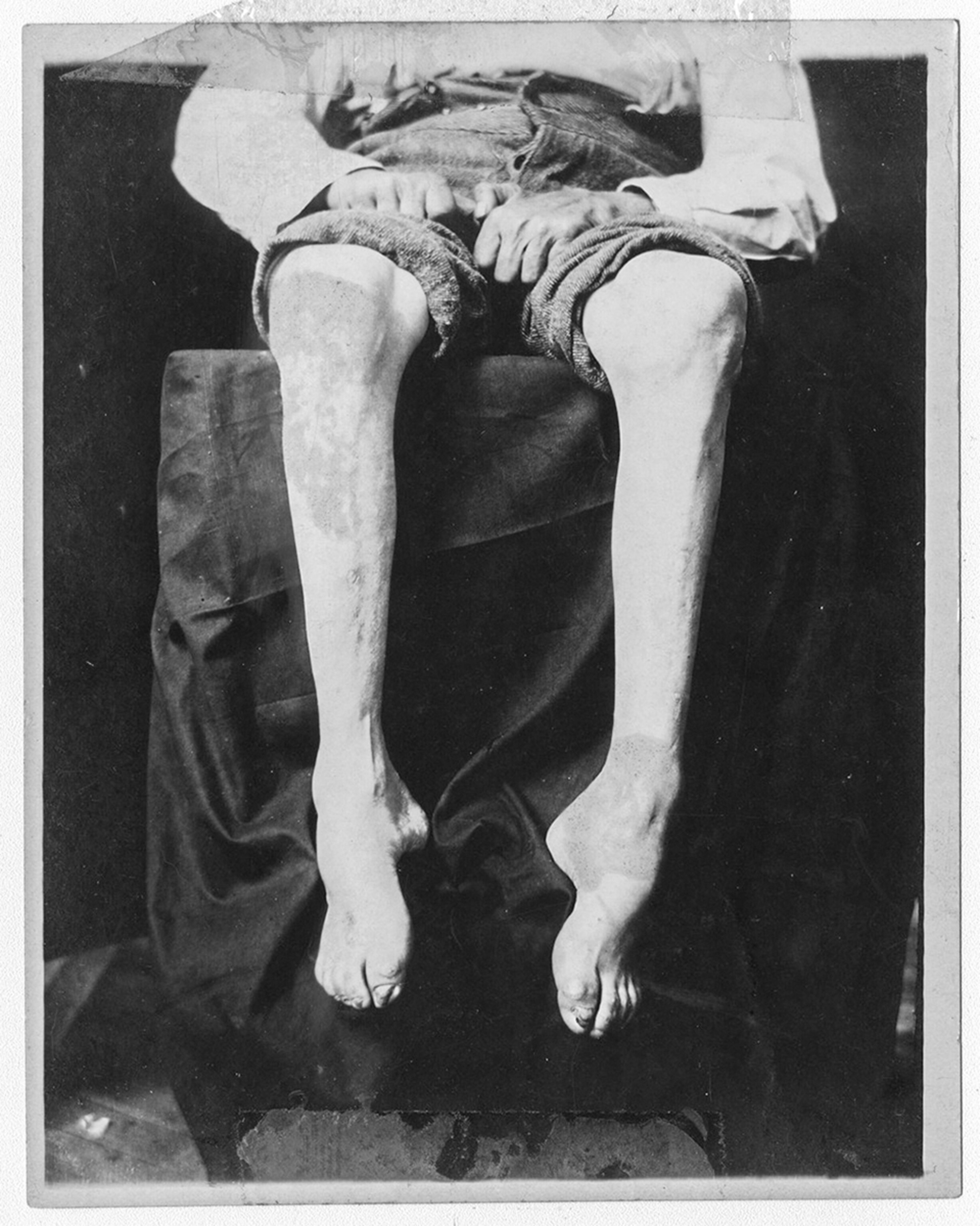
None of this is ancient history. The shift from farm to city began in the industrial revolution, but the human species became predominantly urban for the first time only in 2008. The United Nations estimates that by mid-century, 68 percent of humans will live in urban areas. That means many people once again need to learn that the move from the farm to the city changes how they live. They also need systems that allow them to make that change safely. But many developing nations don’t have the money to pay for sanitary reform.
Today 2.1 billion people lack access to a safe water supply at home, and 4.5 billion lack safely managed sanitation. The absence of both was the main factor sustaining the recent cholera epidemic in Haiti, which sickened at least 800,000 people and killed 10,000 there over nine years. Other victims live in the surging megacities of Asia, Africa, and Latin America. It is as common in large parts of these cities for drinking water to be contaminated with fecal matter as it was for London in 1848, and access to basic medical care is almost nonexistent. So they still suffer old diseases, such as pneumonia, childhood diarrhea, and tuberculosis, which alone killed 1.5 million people in 2018, and also relatively new ones, such as HIV/AIDS, which still kills 770,000 people a year. What’s even more ominous, many of these gigantic cities are close to areas of high wildlife diversity—with an abundant supply of potential new pathogens capable of spilling over to humans. It’s a recipe for breeding new pandemics. Perhaps the ravages of COVID-19, like those of cholera in Chadwick’s London, will become the whip that drives governments to bring sanitary reform to every urban community, as one measure to keep those pandemics from happening.
Late 1800s, Europe: Microbes cause disease
For 200 years, a gathering chorus of voices raised tentative versions of the idea that “animalcules,” or germs, cause disease. Proponents of humoral medicine, and of filth theory, succeeded for a time in shouting them down.
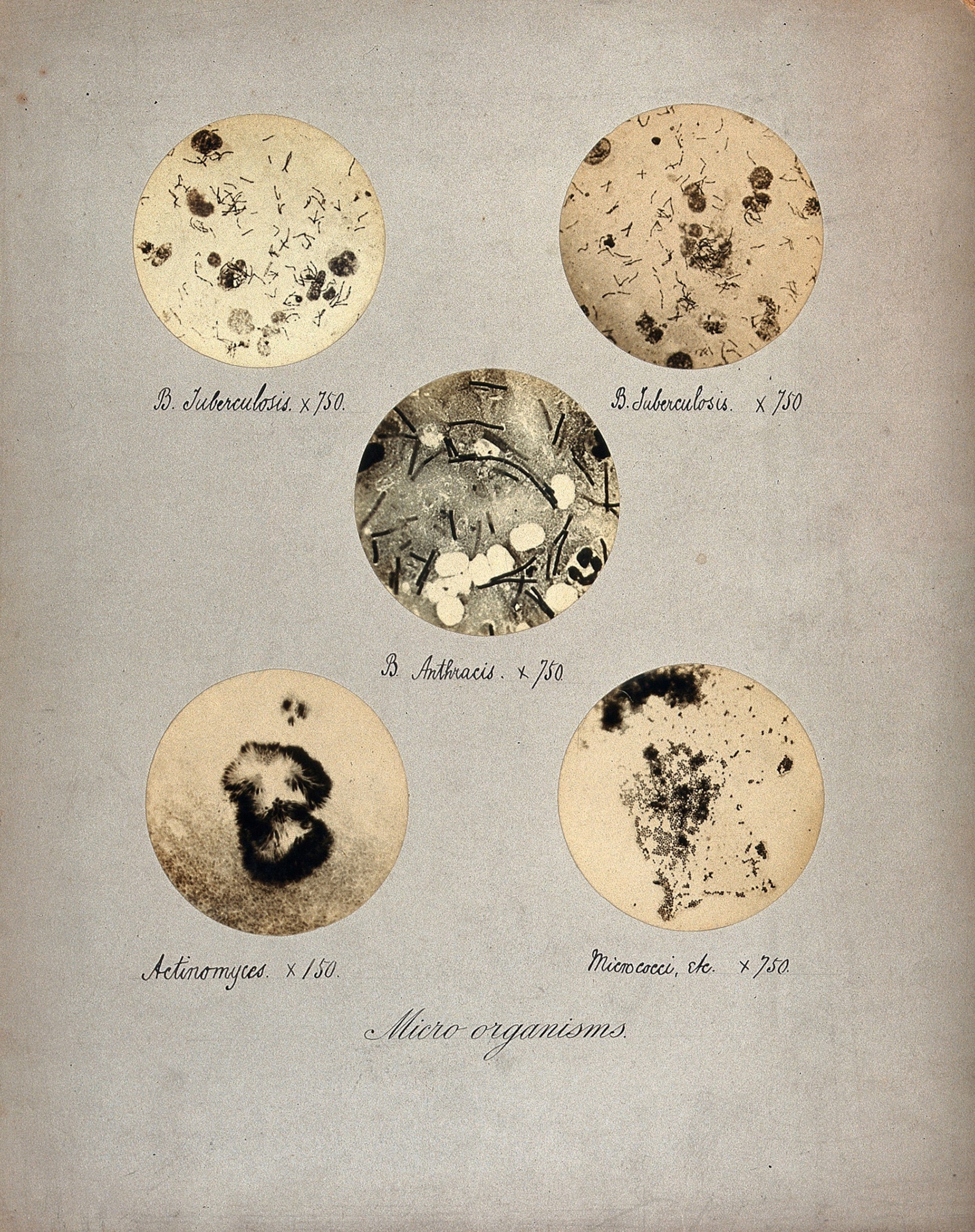
But in the 19th century, as microscopes became more powerful and more widely distributed, other researchers began to open up the world of microorganisms. The idea that specific microorganisms could cause specific infectious diseases—and even decimate human societies—became more persuasive.

History tends now to honor two men, Louis Pasteur and Robert Koch, as the fathers of germ theory and forget the chorus whose work they built on. That’s partly because human nature responds more to a few great names than to the cumulative and collaborative way most discoveries happen. But Pasteur and Koch were also masters of experimental science, meticulous about methods, and brilliant at choosing the right path from one experiment to the next. They hated each other as rivals in the same field of discovery—more human nature—and also as patriots at a time of war between their two nations, France and Germany. But the key breakthroughs they made carried humanity into the miraculous new world of germ theory.
Pasteur was a chemist, not a physician, an outsider perspective that proved useful for bypassing conventional medical beliefs. One of his studies in the 1850s started with the mundane goal of helping a local manufacturer identify the cause of an off taste in batches of beetroot alcohol. Pasteur quickly found the culprit, a type of bacteria, and recommended heating the beet juice to prevent it from happening again—arguably the beginning of pasteurization.
With his characteristic instinct always to look a little further, Pasteur proceeded to detail every stage of fermentation. It wasn’t a purely chemical process, as many “modern” thinkers believed then, but a biological one: Yeast, a living organism, consumed nutrients in the brew and converted them to alcohol and other products. The work on fermentation encouraged Pasteur to see microorganisms everywhere and to demonstrate that they were a product of normal biological reproduction, not spontaneous generation. He went on to make a magnificent intuitive leap: Much as tiny living organisms caused fermentation—and unexpected organisms could spoil a batch—they could also cause infectious diseases.
Pasteur was an enthusiastic self-promoter. (Some modern historians say he also appropriated other people’s work without credit, overstated his evidence, and lied about his methods.) He presented his findings in bold language and marshaled the resources of the French hierarchy in support of his work. He also engaged in fierce attacks on anyone foolish enough to disagree with him about anything, especially germ theory. But it took Robert Koch, then a small-town physician working alone in a home laboratory, to prove that Pasteur’s magnificent intuition was correct.
Koch remains surprisingly little known today; most people are quicker to recognize the name of a minor assistant named Petri who invented a laboratory dish. Twentieth-century anti-German feelings may have turned the hero-making impulse away from Koch. He also lost some admirers when he divorced his wife to marry a beautiful young actress and, at about the same time, promised but failed to deliver a cure for tuberculosis.
Koch deserves better. As a young physician in the mid-1870s in a rural area of what is now Poland, he marked off part of his examining room for a small laboratory. There, between patients, he studied microscopic specimens from the natural world—including blood from a sheep that had died of anthrax. By patient and persistent looking, he gradually unwrapped a hidden mystery about this veterinary disease, which sometimes also kills humans.
Pasteur and Koch hated each other as rivals and as patriots at a time of war between France and Germany. But the breakthroughs they made carried humanity into the miraculous world of germ theory.
Bacteria normally reproduce by dividing in two. In favorable conditions, the repeated doubling of a pathogen such as anthrax can quickly overwhelm a host animal. What no one knew before Koch is that when conditions turn bad, anthrax bacteria can also produce a sort of escape pod. This spore, encased in a tough shell, can survive in the soil in a dormant state for generations, a biological land mine. That suggested an answer to how anthrax sometimes appears out of nowhere, when no new animal has entered a flock, and where no cases of the disease have happened for years or even decades.
Koch soon invented a way to grow the bacteria in an artificial culture, on a piece of glass he could study under his microscope. There he watched the emergence of the spores, and saw them become living bacteria again, with those bacteria subsequently producing a second generation of spores. To show that spores could infect animals after a period of dormancy, he injected them into wild mice—there were no laboratory mice then—quickly giving rise to a new and deadly population of anthrax bacteria.

Koch’s October 1876 paper on anthrax bacteria was a turning point in human history. By repeatedly and predictably producing the symptoms of anthrax in experimental animals, he proved the long-contested reality of contagion and proved that Bacillus anthracis was the agent of that contagion. What he had demonstrated, in short, was the germ theory of disease.
Pasteur and Koch inevitably built on each other’s work, while simultaneously attacking each other in public. Pasteur devised the first new vaccines in the 85 years since Jenner’s smallpox vaccine, including ones for anthrax and rabies. Koch cured no diseases, but he went on to identify the pathogens that cause some of the most terrifying diseases known to humanity, including cholera and tuberculosis, for which he won a 1905 Nobel Prize. He also made many cures possible, by inventing microbiological tools other scientists still use to identify an astonishing rogues’ gallery of deadly pathogens. For the first time, the targeted treatment and prevention of almost any infectious disease became possible.
(Read about researchers' mass production of penicillin, the first effective antibiotic, during WWII.)
Present day
By the time infectious disease specialist Michael Callahan climbed aboard the Grand Princess in early March, he was already an old hand in the COVID-19 pandemic. He’d gotten his start in January trading notes in his tightly knit network of fellow experts about the emerging pathogen in Wuhan, China. He saw patients in Singapore as the disease was breaking out there, and he briefed U.S. government officials in Washington, D.C., on where it might occur next. He helped evacuate a cruise ship in Yokohama, Japan.
Then he treated early victims as the disease spread to Boston, where he is a staff physician at Massachusetts General Hospital. And as he watched and worked, and brainstormed ventilator issues, he saw the disease reveal its “magnificent infectivity,” its ability to sit “like a little silent smart bomb in your community,” till it finds a person “and just takes them out.”

“When I saw my 500th patient, I became terrified,” Callahan says. “It’s a sleeper.”
For decades Callahan has been a familiar face on the front line of epidemics everywhere, working to end outbreaks of Ebola, SARS, H5N1, and a deadly alphabet soup of others. He fits his own description of experts who turn up on the scene of new outbreaks: “high-strung, fast-moving, compressed-speech people.” Answering questions by phone for this story, he delivered lengthy, complex answers in high-energy bursts, with a dizzying tendency to hopscotch across the medical and geographical world.
But even among his highly skilled, hypermotivated colleagues, Callahan stands out for his ability to synthesize information in a crisis to quickly get to the best available option. As such, he’s on speed dial for an array of organizations, from hospitals and global health nonprofits to the U.S. government, where he’s the special adviser for COVID to the assistant secretary of preparedness and response. Now and then, he also gets home to his family in Colorado, where he works by phone and laptop, interrupted, like everyone else in mid-lockdown, by the dog barking, a child in need of bicycle repair, and the eternal call to neglected household chores.
Callahan chose this career path because of a brutal stint at refugee camps in eastern Democratic Republic of the Congo in the late 1990s. It taught him that infectious disease in the developing world is a “slow-rolling disaster. And it goes on forever. I became very driven by the unfairness of it all.”
We have entered a frightening new world. Or maybe we are returning to the old world of our disease-plagued ancestors. Either way, the one great lesson we should take away is not to forget this happened.
His subsequent experience with Ebola and other outbreaks in West Africa also taught him that treating one case at a time wasn’t enough. Rather, providing training or supplies to local medical staff “would lead to huge changes in a village or community or hospital. And those changes would endure after you left.”
That became his guiding philosophy. Working for a program run by the U.S. State Department, he helped physicians and scientists displaced from chemical and biological weapons programs in post-Soviet Russia retrain to become peacetime infectious disease researchers. That led to almost a decade at DARPA (the Pentagon’s Defense Advanced Research Projects Agency). There he developed a program called Prophecy to predict and preempt emerging diseases.
Callahan’s background has given him unusual insight into how we might adapt to COVID-19, and other emerging diseases that lie beyond. Protecting our health, he suggests, can depend on finding ways to help other countries meet their own needs, even if the national government is outwardly hostile and those needs might not always seem to serve our short-term national interests. It’s about playing a longer game.
In Indonesia, for example, overfishing has decimated coastal seafood stocks, and Islamic law prohibits eating pork. That makes maintaining a secure protein supply a major issue—especially after an outbreak of avian flu caused severe losses to the poultry business. So the Prophecy program at first quietly focused on protecting chicken stocks. Among other measures, it provided local gene-sequencing capability that allowed Indonesia to identify pathogens on its own, reducing dependence on Western powers.
It was the sort of initiative that might easily attract scrutiny from Congress, or from isolation-minded critics. But in Indonesia, “our equity went up,” says Callahan, “and we got into the good stuff,” meaning human pathogen surveillance. “DARPA, an otherwise secretive military agency, was a welcome partner.”
Prophecy’s other important strategy was to find smart young infectious disease physicians in developing countries and build lifelong relationships. That could mean supplying them with new technology, bringing them to U.S. medical schools for further training, or providing grants for new research.

“And by promoting the foreign partner, they get promoted themselves, and they become leaders in their field, and two things happen,” Callahan says. “They develop host-nation sustainable funding. And you have a very grateful emissary who’s now at the pinnacle of … pathogen intelligence.”
One such foreign partner was a researcher in Russia Callahan had helped make the transition from bioweapons to disease detection. In 2005 that researcher’s laboratory spotted an outbreak of H5N1, an avian flu that can devastate poultry and wild birds. It’s also capable of jumping to humans and killing young people by destroying their lungs. It was moving northeast to where Asian and American flyways overlap in the Bering Strait region. The early alert enabled U.S. scientists to launch a major testing program on migratory birds in Alaska and prevent the disease from entering the continent.
The Prophecy program expired a few years after Callahan’s time at DARPA ended. (DARPA’s mission, says Callahan, is to invent new programs, not manage them. But some of the tools Prophecy introduced have gone on to help in quickly developing new vaccines and in predicting when diseases will become resistant to antimicrobial drugs.) The larger tendency among governments everywhere has been to discount the risk of pandemics and to underfund programs designed to prevent them. Thus, late last October, the U.S. government allowed Predict, another program focused on emerging diseases, to end. Less than a month later, the first known COVID-19 case occurred in China. And soon after, American victims began joining the worldwide ranks of the dead.
The current pandemic will almost certainly step up efforts to predict and control pandemic diseases, at least for a time. But no one knows yet what shape prevention should take, what it will cost, or how devastated economies will pay for it.
Will nations play the long game of international cooperation? Or will the trend to short-term national self-interest become more pronounced? Will a society that has barely quibbled about spending $13 billion on an aircraft carrier, largely in the service of preventing armed conflict, also accept spending on an even grander scale to prevent epidemic diseases? Will we continue to spend indefinitely, even though this kind of prevention means having nothing tangible to show for our money, no heroic physical object, just the unsatisfying knowledge that the catastrophe we feared did not happen?
We have entered a frightening new world. Or maybe we are returning to the old world of our disease-plagued ancestors. The one great lesson we should take away from history is this: When the current pandemic ultimately subsides, we cannot afford to forget that this happened. We cannot just move on. Somewhere on the planet, the next great pandemic, the next destroying angel, is already taking wing.