
Could Fido be our best hope to reverse aging in humans?
A canine elixir for longevity could lead to one for us: They live in our houses, breathe the same air, and suffer many of the same ailments that affect older people.
Stephanie Abraham, a dog breeder and dog-show judge in Connecticut, lives with eight laid-back Cavalier King Charles Spaniels and a mischievous one named Ace. “He’s my only Cavalier who gets into trouble!” she says. The nine-year-old 18-pounder, with rich chestnut-color markings and large, soulful eyes, likes to leap on a kitchen chair, swipe mail off the butcher-block counter, and munch it to shreds. “The latest thing he chewed was my summons for jury duty,” Abraham says.
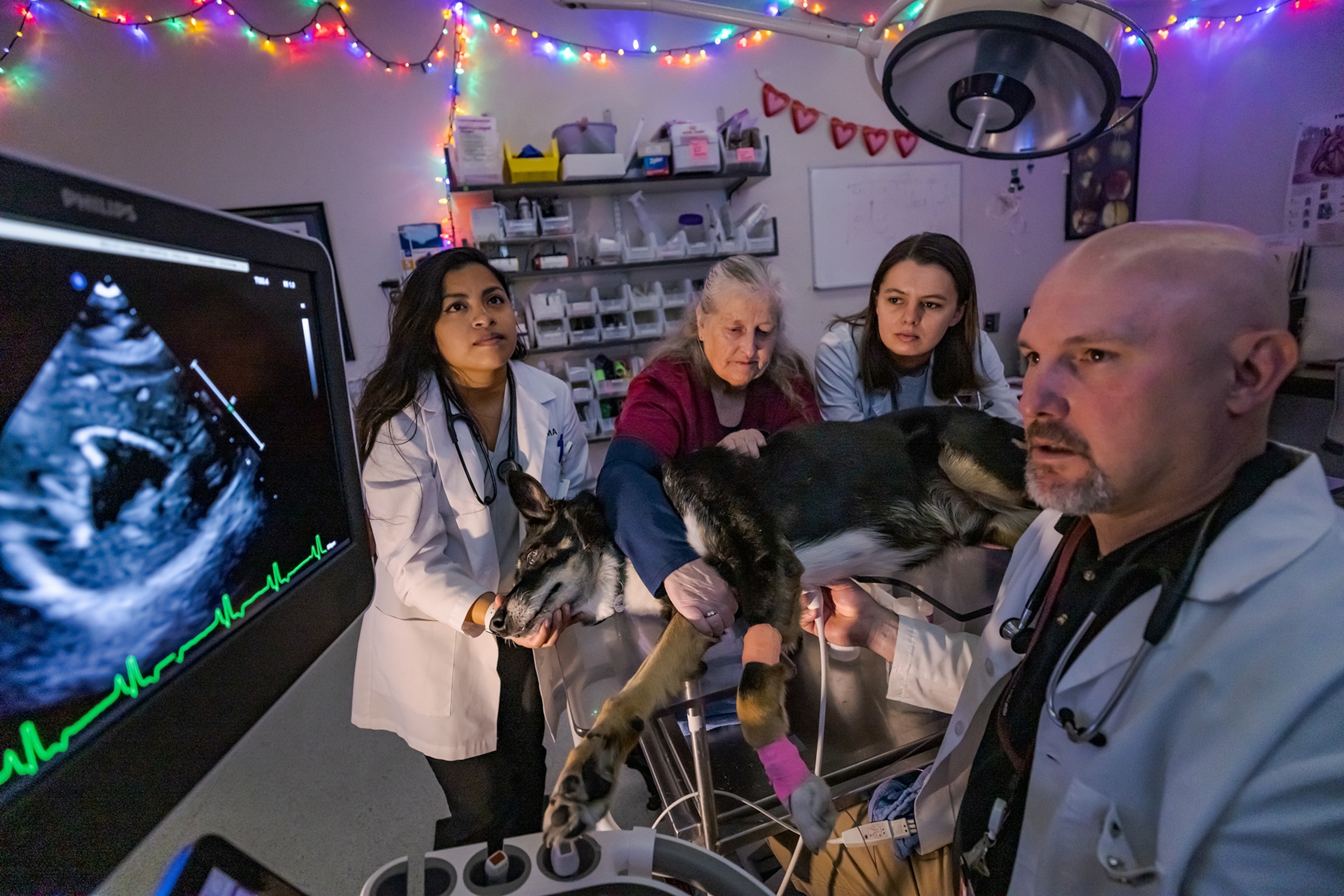
A few years ago, Ace, an American Kennel Club champion, developed a mild heart murmur, the first hint of a progressive valve disease that can eventually lead to heart failure and death. It affects about 7 percent of all dogs and as many as 80 percent of Cavaliers. Drugs may slow it down, but Ace’s murmur worsened. So Abraham signed him up for an experimental treatment, one that may usher in a new generation of gene therapies to cure chronic diseases and maybe aging itself—not only in dogs but in us.

The treatment Ace received, code-named RJB-01, delivers two genes associated with longevity. Most gene therapies take aim at a single gene defect—for example, an inherited form of blindness—but this treatment doesn’t target a mutation or even the faulty valve. Instead, it tries to restore cellular processes and pathways that are important for health but break down with age, leading to heart problems, diabetes, and other chronic ailments.
“We view the world like this—we think aging is reversible,” says Daniel Oliver, co-founder and CEO of Rejuvenate Bio, the gene therapy’s developer. “If you are able to affect aging, you should be able to affect multiple age-related conditions.”
For decades, scientists trying to push the limits of human life span have studied simple organisms like worms and fruit flies, and those laboratory mainstays: mice and rats. Various drugs and calorie-restricted diets have been shown to delay aging and prolong life in these species, sometimes by as much as 50 percent. But the experiments haven’t led to a breakthrough for us, and though it may sound cold-hearted, Oliver notes that the world isn’t waiting breathlessly for longevity drugs for worms and rats. “Most people don’t care about extending their life span,” he says.
By setting their sights on dogs, longevity researchers hope to discover ways to turn back time in humans. Dogs make good models for investigating why we decline, physically and cognitively, as we age. They share our households, breathe the same air, often chow down the same foods, and suffer many ailments that plague older people, including cancers, arthritis, diabetes, dementia, obesity, frailty, and like Ace, mitral valve disease. Because a dog’s life zooms by much faster than ours, scientists can track biological changes in a matter of years, not decades, and test anti-aging therapies at far less cost.
Even if experiments in dogs yield nothing to thwart human aging, they may produce novel treatments for our furry friends—and nobody who has ever bid farewell to a cherished canine would object to that. “If we solve the problem of longevity with dogs, we will reduce lots of grieving and suffering,” says molecular biologist and cancer researcher Andrei Gudkov, who co-founded an organization that’s studying aging in retired sled dogs. He named it Vaika, after his late Siberian Husky.
Of course, the motivation for biotech startups isn’t purely humanitarian. Americans have about 77 million dogs, making them the most popular pet by far. Some ardent—and affluent—owners have spent upwards of $50,000 to clone an adored pooch. Riches await the company that unleashes a longevity elixir for Fido, whether or not it leads to one for us.

Canines on the cutting edge
Biologist Matt Kaeberlein at the University of Washington co-directs the Dog Aging Project, the most ambitious effort to try to crack the code on canine longevity and glean the secrets it holds for humans. Started in 2019, the project has enrolled nearly 40,000 pet dogs of all kinds in a 10-year study to identify the biological, environmental, and genetic factors that promote healthy longevity.
Their owners submit medical histories as well as detailed surveys on the health, cognition, and “life experiences” of the dogs. The researchers also collect information on air and water quality in the zip codes where their subjects live. Ten thousand of the dogs are slated to have their genome sequenced. About a thousand will undergo extensive veterinary exams annually, with sophisticated analyses of their microbiome and epigenome, or chemical modifications on DNA that indicate biological age.
Scientists have already published a slew of papers based on the project’s data, including two recent ones with conclusions relevant to people: Physical activity protects against cognitive dysfunction and one daily meal is better than frequent feeding for all-around health.
Kaeberlein is also leading a trial of rapamycin, an immunosuppressant approved by the U.S. Food and Drug Administration for organ transplant patients, in 580 dogs. Dozens of studies have shown it extends the lives of mice and other model organisms.
Some scientists consider rapamycin one of the most promising candidates for a human longevity drug. More than a few people take it off-label for that purpose. Kaeberlein has put out the word that he’d like them to send him information on their health. He also pops a low-dose rapamycin pill weekly, in intermittent 10-week-cycles, and is beginning to monitor the effects on his blood biomarkers and epigenome.
The drug triggers some of the same molecular and metabolic action as extreme calorie restriction, which has consistently extended life span in laboratory studies. Kaeberlein believes rapamycin may also regulate inflammation, an important factor in age-related disease.
But self-experimentation and crowd-sourced anecdotes won’t prove anything, and rapamycin is off-patent, so the pharmaceutical industry has little incentive to fund a large clinical trial. “There is no money to be made, or at least not as much money as if it was a new drug,” Kaeberlein says.
His double-blind, placebo-controlled dog study is enrolling healthy seven-year-olds, so a significant gain in life-extension would become apparent in three years. Depending on his findings, they could shake loose money for clinical trials or consign rapamycin to the long list of drugs that initially raised hopes for a longevity breakthrough but fizzled.
Andrei Gudkov believes there are too many variables in pet households to provide clean scientific data. So he and his colleagues in the Vaika study recruited 102 former sled dogs, ages eight to 11, from across the U.S. to live out their golden years in a Cornell College of Veterinary Medicine kennel under tightly controlled conditions, with ample space to run and play.
The scientists scrupulously monitor the changes of later life—the dogs do treadmill tests, cognitive tasks, and problem-solving activities such as figuring out how to get around a fence. The scientists also have tested two drugs for their anti-aging potential: lamivudine, an FDA-approved treatment for HIV and hepatitis B, and entolimod. A recombinant protein developed to counter the effects of radiation poisoning, entolimod is also being evaluated at the Mayo Clinic as an immune-system booster in people 65 and older.
Meanwhile, a San Francisco startup with the canine-reverent name of Loyal is testing a dissolvable implant that releases a drug aimed at slowing aging in large breeds, which grow old faster and die younger than small ones. Also in the pipeline: a tasty pill for older dogs of all but the tiniest toy breeds, code-named LOY-002. Like rapamycin, it emulates the biological effects of calorie restriction.
“While we are building these dog longevity drugs and giving something that pet parents, at least from the emails I get, really, really want, we're also learning something about how to help people live longer, healthier lives,” says Celine Halioua, Loyal’s founder and CEO. “Honestly, the most important thing that Loyal can do is prove that aging should be a drug class … that there's a way to develop a drug for this mechanism.”
The DNA cure
Ace’s mother, Gabby, developed mitral valve disease before he did. The leaky valve causes blood to flow back into the upper left chamber of the heart instead of moving to the lower chamber. At 12, Gabby was part of the first pack of dogs to receive the gene therapy that Ace would later get. Their treatment grew out of experiments led by Noah Davidson, then a postdoctoral researcher in the lab of Harvard biologist George Church.
Davidson knew that gene expression—the process by which information stored in DNA is translated into molecules that control how cells function—can go awry as we grow older. He believed that properly regulating gene expression, which means switching on some genes and turning off others, was the key to slowing aging and eliminating many diseases that come with it.
He and his colleagues zeroed in on three genes known to promote healthy aging and longer life in genetically engineered mice. He theorized that an extra copy of any of these genes, or maybe all of them, would have broad health benefits in normal mice. The team created a therapy from each gene and tested them all in mice, one therapy at a time and in two- and three-gene cocktails. In a 2019 paper in PNAS, the scientists reported that a single dose of a two-gene combo mitigated four age-related ailments: type 2 diabetes, obesity, heart failure, and kidney failure.
Rejuvenate Bio, co-founded by Church, Davidson and Daniel Oliver, quickly jumped to tests on dogs. The study, focused on evaluating the therapy’s safety, isn’t limited to Cavalier King Charles Spaniels. But a passionate, well-organized community of owners of the breed, most of whom will have a dog with mitral valve disease, spread word of the trial. “This is a very big deal in the Cavalier world,” Stephanie Abraham says.
In early 2020, Abraham drove Gabby to the Cummings School of Veterinary Medicine at Tufts University, in Massachusetts, an hour from their home. The dog received an intravenous infusion in her hind leg. For all the eye-popping science and painstaking research that went into creating the therapy, the IV drip took less than 15 minutes. “There was no pain or crying,” Abraham says. Ace breezed through the infusion two years later.
Rejuvenate Bio has not announced the results, but it has partnered with an animal health company and plans to seek FDA approval for the canine gene therapy. The startup also plans to recode the gene cocktail for human use and test it for two ailments: arrhythmogenic right ventricular cardiomyopathy, which breaks down part of the heart’s muscular wall and increases the risk of irregular heartbeat and sudden death; and familial partial lipodystrophy, a disorder of abnormal fat storage that leads to diabetes, an enlarged liver, and other health problems in adulthood.
Gabby and Ace have had no discernible complications or side effects from the therapy, and Abraham is encouraged by blood tests for a hormone that indicates how well the heart is pumping blood and can flag incipient heart failure. Gabby’s levels have improved; Ace’s are stable. The scamp shows no sign of keeping his paws off the mail.