‘Microclots’ could help solve the long COVID puzzle
Scientists are starting to detect clots in long COVID patients’ smallest blood vessels—which might help explain the condition’s debilitating symptoms.

For more than two years, scientists have been trying to understand why millions of people across the world are experiencing lingering symptoms despite recovering from their COVID-19 infection. They’ve proposed several hypotheses including the presence of microclots—tiny blood clots that can block capillaries and potentially affect blood and oxygen flow.
In a 2021 study, physiologist Etheresia Pretorius at the Stellenbosch University in South Africa and her colleagues were the first to suggest that microclots may be linked to this debilitating condition called long COVID. In a follow-up study, she and her colleagues showed that the SARS-CoV-2 spike protein triggers the formation of such clots, which the body’s natural clot-busting process doesn’t seem to break down easily.
This finding has led some scientists in the United States, with guidance from Pretorius, to test people with long COVID for microclots. Lisa McCorkell, co-founder of the long COVID-focused Patient-Led Research Collaborative, was thrilled when she heard the news last year.
McCorkell had experienced severe shortness of breath, extreme fatigue, and brain fog for several months following her mild COVID-19 symptoms in March 2020, when the pandemic began. In August that year, when she started to feel better, McCorkell took a workout class. But a day later, her heart rate spiked, she struggled to breathe, and she rushed to the emergency room. “That lowered my baseline quite a bit,” she says. “Before COVID, I was running half marathons, so it was a very dramatic change.”
In December 2020, the 28-year-old finally came to terms with how sick she was and that her illness wasn’t temporary. In late 2021, her suspicions were confirmed when she was diagnosed with postural orthostatic tachycardia syndrome (POTS), a condition documented in several long COVID patients that can disrupt breathing and cause heart palpitations and dizziness on standing up. POTS has no cure and some patients, including McCorkell, manage symptoms by increasing fluids and salt intake. But a year after her diagnosis she still suffers post-exertional malaise that worsens these symptoms.
What’s frustrating for McCorkell and many other long COVID patients is blood and other routine tests turn up normal despite their debilitating condition. In November 2022, she flew from California to New York where David Putrino, a rehabilitation and long COVID scientist at Mount Sinai Health System, and his collaborators are collecting blood samples to search for microclots. “We’re very early,” Putrino says. “We’ve only tested a few dozen folks so far.” But every sample from long COVID patients, including McCorkell’s, has revealed such clots.
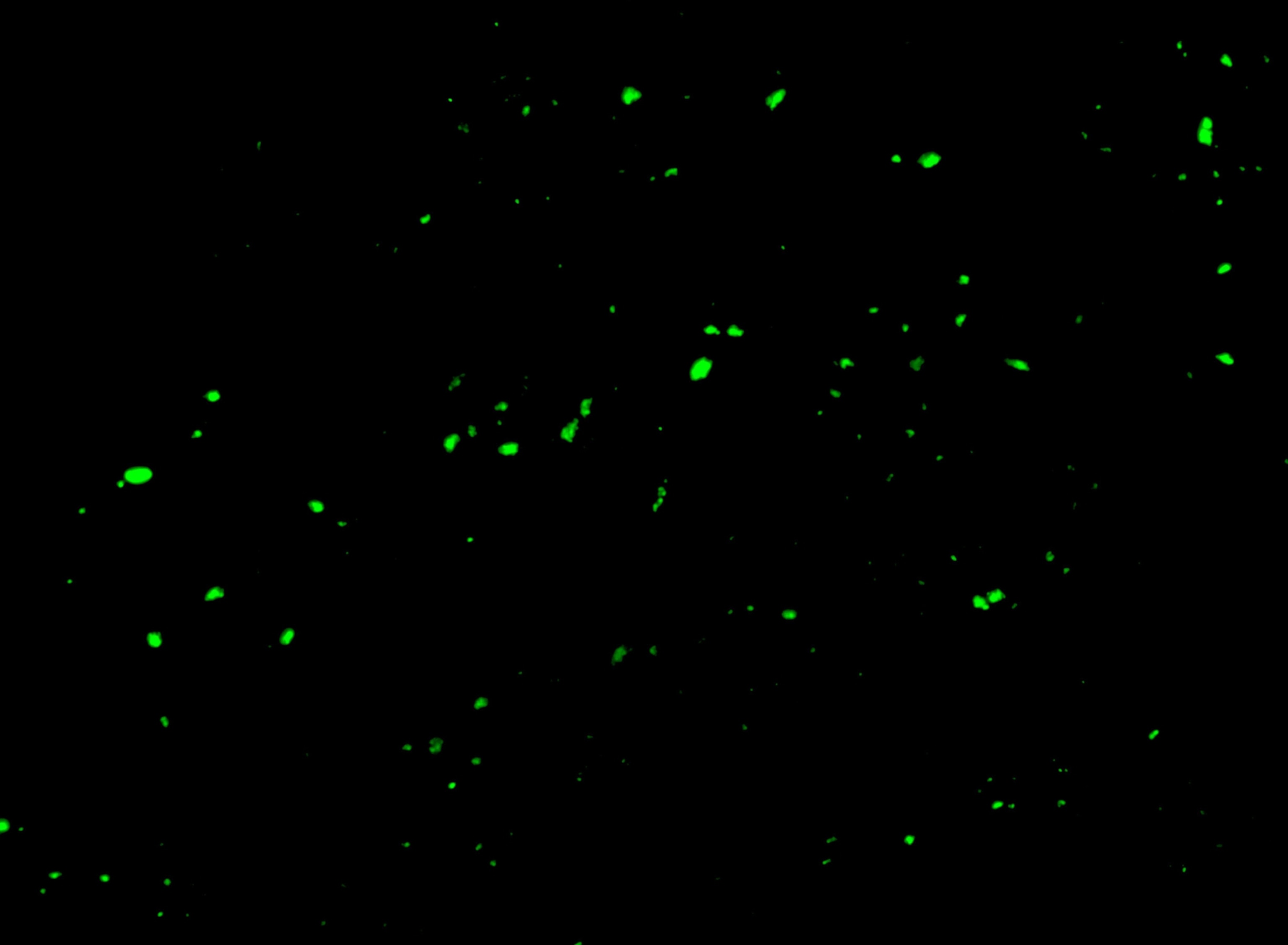
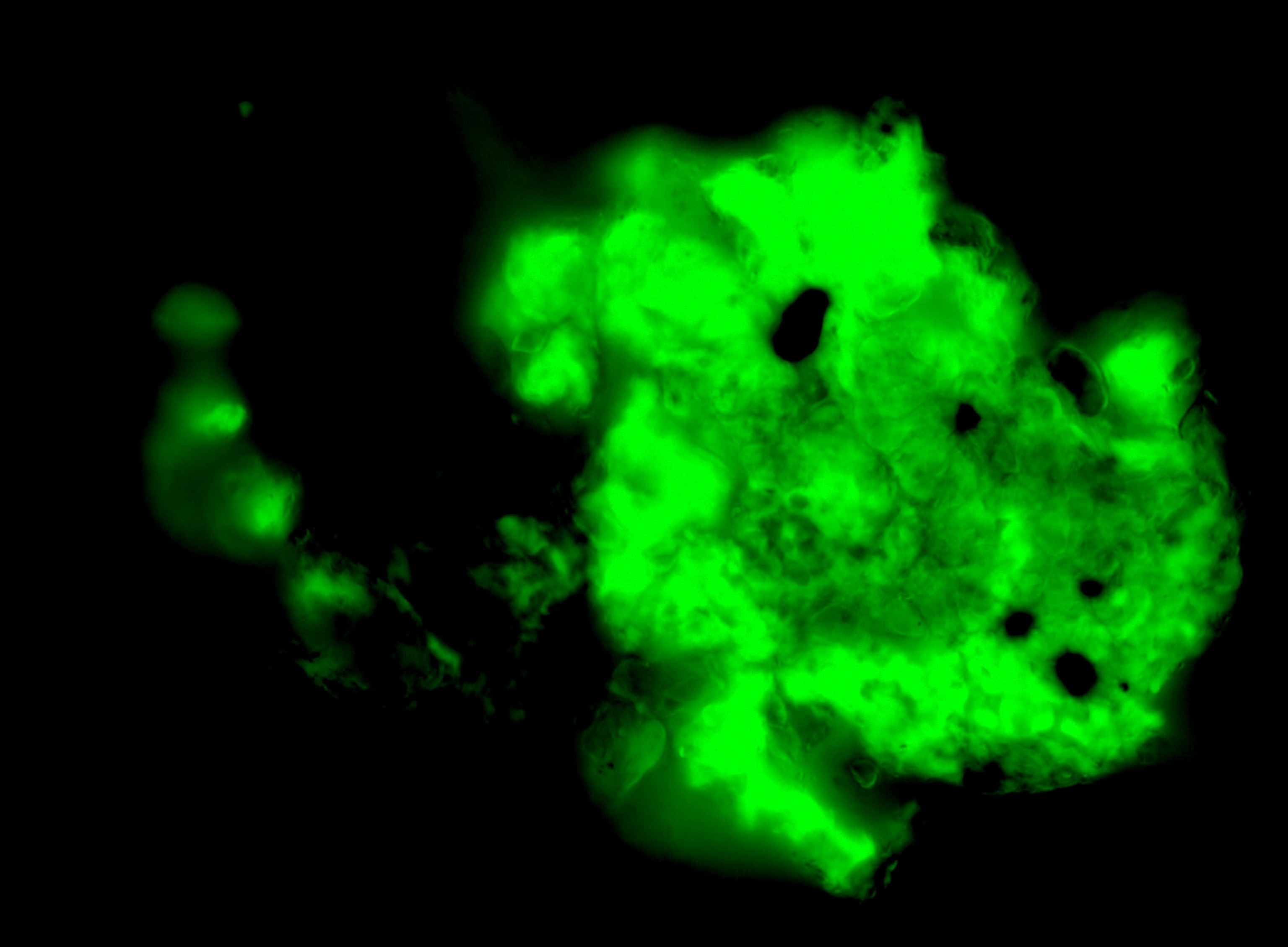
When she first saw the microscope images of fluorescent green blobs revealing the microclots, she cried with relief. For her, the confirmation that she has microclots felt like validation of her illness, “especially after not getting a PCR test at the beginning and being gaslit throughout the last few years.”
While some experts agree the microclots hypothesis is plausible, they think it could be just one piece of the long COVID puzzle. But they want to see more research that demonstrates how these clots contribute to long COVID symptoms and whether getting rid of them leads to improved health outcomes.
How microclots form
Unlike blood clots that block arteries or veins, microclots occur in small blood vessels. They form when a soluble protein called fibrinogen is exposed to inflammation-causing molecules, which can bind to the fibrinogen and aggregate into sticky blobs. “They are not capable of clogging large vessels; they’re not capable of causing life-threatening symptoms,” Putrino says, but notes, “They can significantly affect organ function.”
Pretorius and her colleagues have been studying such microclots for more than a decade and have observed them in patients with type 2 diabetes, chronic fatigue syndrome, Alzheimer’s, and Parkinson’s disease. In a preliminary 2021 study, they saw substantial microclot formation in the blood of acute COVID-19 patients, as well as people with long COVID who experience persistent symptoms for six months or longer. “The main difference between microclots we find in diabetes and other conditions is that they break up quite easily,” Pretorius says. COVID microclots are harder to disintegrate.
Trapped inside the persistent microclots, her team found high levels of inflammatory molecules and a protein called alpha 2-antiplasmin that prevents their breakdown. Such blockages in tiny blood vessels throughout the body could hinder the supply of oxygen and nutrients to the organs and tissues, potentially leading to long COVID symptoms like fatigue, muscle pain, and brain fog.
But what’s triggering the microclots formation? Pretorius and her colleagues think it’s the SARS-CoV-2 spike protein, which can linger in the blood of long COVID patients for up to a year. In a 2021 study, the team added spike proteins to healthy blood and were able to trigger the development of microclots. They also found that in the presence of the spike, the microclots were more resistant to fibrinolysis—a natural process that enables the removal of clots. “Our belief is that the spike protein binds to the healthy fibrinogen,” Pretorius says. “We think that interaction perhaps makes for a tighter [microclot] structure and a bigger structure.”
If these microclots persist for prolonged periods, the body could produce autoantibodies—proteins that inadvertently attack the body’s own healthy tissues and cause debilitating disorders. “It’s those individuals who we are particularly worried about,” she says.
How scientists detect microclots
Detecting microclots requires a specialized laboratory technique called fluorescence microscopy. “You can’t just go to the doctor’s office and get tested for microclots,” says microbiologist Amy Proal, of the nonprofit PolyBio Research Foundation and co-founder of the long COVID Research Initiative.
The process involves drawing blood, spinning it, and adding a fluorescent agent to see the clots under a fluorescence microscope. It’s not a widely available tool in general pathology labs.
But what’s unknown is the sensitivity and specificity of this method. “If you’ve got 500 long COVID patients, is this assay positive 100 percent of the times or 20 percent,” asks hematologist Jeffery Laurence at the Weill Cornell Medical College in New York City, who isn’t involved in Putrino’s or Pretorius’s research. “Given that a similar phenomenon occurs in other diseases, how specific is this for COVID.”
He also points out that published microclot studies have been done in a small number of long COVID patients, but future work should involve testing blood samples from many more people and replicating the research in several labs. Putrino, in collaboration with immunologist Akiko Iwasaki, at Yale University, plans to test hundreds of long COVID patients “because a few dozens is by no means valid for saying everybody [with long COVID] has microclots,” he says.
For now, Putrino and his team are seeing a correlation between the number of microclots on a microscope slide and the severity of a patient’s cognitive impairment. These include their ability to regulate emotions, plan and put together long-term solutions to problems, or figure out ways to deal with real-time situations as they’re changing. The research team is also developing an objective measure for microclots. “We’re still at a very rudimentary stage,” Putrino says.
Hematologist Yazan Abou-Ismail at the University of Utah, who isn’t associated with the microclots research but finds the theory plausible in the context of long COVID, also hopes to see studies that document what’s happening inside the capillaries and organs of long COVID patients with microclots. “It can be hypothesized that the microclots end up obstructing small blood vessels,” he says, “but we don’t really know whether there’s an actual obstruction.”
Treating microclots
While researchers try to determine the prevalence of microclots in people with long COVID and why they form, patients are suffering and desperate for treatments.
In a December 2021 preprint study, which is yet to peer-reviewed, Pretorius and her team showed a decrease in microclots and reduced platelet activation—a condition that accompanies microclot presence—in 24 long COVID patients who were administered a combination of anticoagulant Apixiban and a dual antiplatelet therapy for a month. However, they’re in the process of revising the study to include more patients and measurements of their health outcomes following the treatment. “But we need clinical trials to show anticoagulation approaches and antiplatelet approaches have efficacy,” Putrino says. He also wonders if clots in small blood vessels may need different anticoagulants compared to those used against large clots.
McCorkell, on the other hand, is taking her treatment into her own hands and experimenting with over-the-counter enzyme supplements like serrapeptas and nattokinase that seem to breakdown blood clots but aren’t approved by the U.S. Food and Drug Administration.
Like many other people with long COVID, McCorkell is disappointed and angered that there aren’t clinical trials to test the use of such supplements and other off-label therapies that some patients are resorting to for relief. Many health providers are also often unable to help. Although she hasn’t experienced any side effects so far, McCorkell knows of some individuals who have had nausea and vomiting episodes from taking the same supplements. Pretorius and her team plan to conduct a study to test if these supplements are effective, but until then many patients are on their own.
“Given the scale of the issue and how much it impacts people’s lives, we need an Operation Warp Speed situation,” McCorkell says. “It’s frustrating that we’re not further along.”