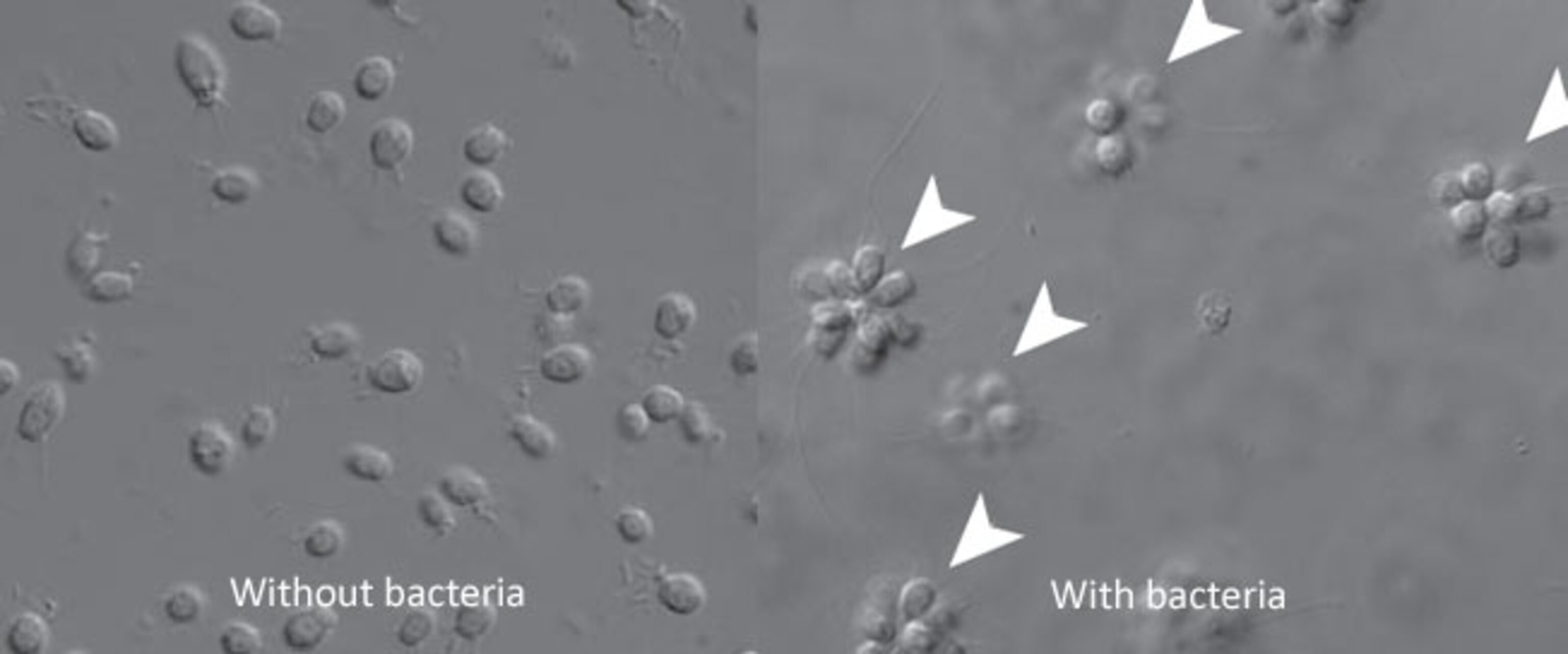
Bacteria transform the closest living relatives of animals from single cells into colonies
In Nicole King’s lab, a bacterium is making a group of tiny cells stick together. That might seem a little humdrum for a group whose members can build electric grids, create snow, and cripple nations. But King’s bacteria should not be overlooked, for they are recapping one of the most important events in the history of life: the move from one cell to many.
The cells in question are choanoflagellates – the closest living relatives of all animals. They’re not our direct ancestors, but they give us clues about what those ancestors were like when they were still swimming around as single cells. Choanoflagellates normally live in solitude, moving about with sperm-like tails and voraciously eating bacteria. But they can also form big colonies. If we can understand why this happens, we might get hints as to why our single-celled ancestors did the same.
King has now found the answer, and it’s a tantalising one. The solitary cells become sociable after being exposed a molecule called RIF-1 that’s produced by some of the bacteria that they eat. When they divide in two, the daughters normally go their separate ways; add a splash of RIF-1, and they stick together instead.
This raises an obvious question: did bacteria also help the single-celled ancestors of animals to band together? Did they contribute to the evolutionary foundation of every ant and elephant, every fish and finch? “That’s my favourite hypothesis,” says Rosie Alegado, the lead author on the new study. “Animals evolved in seas teaming with bacteria and have been passively exposed to bacterial chemical cues, intended and unintended.” But she cautions that this is still an open question.
Her research has certainly excited Michael Hadfield from the University of Hawaii at Manoa, who studies the development of animals. “This discovery is one of the most exciting for evolutionary biology in many years,” he says. In his view, King’s bacteria are inducing the creation of a primitive embryo and “hinting at a very important bacterial role in the evolution of animals.”
Hadfield also wonders about the connection to sponges. These were some of the earliest animals to appear, and they have cells called choanocytes that look remarkably like choanoflagellates. They have the same basic shape, and they use their beating tails to create currents that sweep food into the sponges. Hadfield says that sponges are hosts to dozens of bacterial species, which form dense swarms around sponge embryos. “For me, this raises the question: are the bacteria essential for the cohesion of cells in sponge embryos?” he says.
William Ratcliff, who studies the origins of many-celled life, has a more measured take. “This paper shows that environment really matters,” he says. The choanoflagellates fundamentally change how they grow when one of the species they eat is around. “Nobody would have predicted this, and it should serve as a reminder to be cautious about inference from lab studies in artificial conditions, where “unknown unknowns” may have a large effect.”
But he is less convinced that the results are relevant to the origin of multicellular (many-celled) life. “Modern day choanoflagellates are not representative of the ancestors of animals,” he says. They’ve been evolving on their own for more than 600 million years, and “they have diverged from animals in significant and largely unknown ways”. As such, we can’t assume that a relationship between modern choanoflagellates and modern bacteria reflects what happened when the single-celled ancestors of animals started bandying together.
This new discovery is the culmination of years of frustration, hard work and serendipity. When King first started studying choanoflagellates in 2000, many people had seen these creatures form colonies in the wild, but no one could duplicate those colonies in a lab. She became the first, after a trip to Virginia. There, a colleague Tom Nerad found beautiful choanoflagellate colonies in coastal water samples.
King is still working with the same choanoflagellates today – a species called Salpingoeca rosetta. It hasn’t been easy. In the wild, the species forms colonies galore. In the lab, “it made colonies only rarely and unpredictably,” King recalls. Her team tweaked every conceivable parameter in their quest to nudge the single cells into flourishing colonies, but to no avail.
Frustrated, King turned to a different goal: sequencing the genome of S.rosetta. That’s when her luck started changing. To grow S.rosetta, you need to feed them with bacteria, but bacteria can also contaminate the results of sequencing efforts with their own genes. To get rid of them, undergraduate Richard Zuzow fed the choanoflagellates with different antibiotics, but ended up disrupting the ability to form colonies entirely. Zuzow took bacteria from the original coastal samples that Nerad collected, and added them back to the choanoflagellates. One bacterium induced the growth of colonies.
Alegado took over. She identified the species responsible – a new one called Algoriphagus machipongonensis – and showed that its relatives can also turn the choanoflagellates towards colony life. This group, known as Bacteroidetes, have a long association with animals. For example, it’s one of the groups found in our guts, and it influences how skinny or fat we are.
Alegado also purified the molecule responsible for the bacterium’s ability, with help from Jon Clardy’s team of chemists at Harvard Medical School. The substance, known as RIF-1 (rosette-inducing factor) is a fat-like molecule called a sulfonolipid. On its own, RIF-1 induces rosettes and at extremely low concentrations –just a few thousand molecules in a millilitre will do. At such infinitesimal concentrations, it took Clardy’s team over two years to isolate enough of RIF-1 to identify it in the first place!
Alegado writes that S.rosetta might be using RIF-1 as a sign of how many bacteria there are around it. Colonies are better at capturing bacteria than individual cells, so if RIF-1 is abundant, it makes sense for the choanoflagellates to band together to better exploit the feast of riches. Ratcliff says that another alternative is that the bacterial chemicals are so widespread in natural bodies of water that “choanoflagellates have lost the ability to produce them, and don’t develop properly when they are absent”.
There’s still plenty to discover. Does S.rosetta need to actually eat the bacteria to respond to RIF-1, or can it pick up the molecule from a distance? What does RIF-1 actually do in the choanoflagellates to trigger the colony lifestyle? Does RIF-1 affect animals today? And for that matter, did it affect our ancestors in the past?
“Do I believe that a RIF-1-like molecule contributed to animal origins?” asks Alegado “I donʻt know. If I am allowed to delve into rampant speculation, I would say that bacteria in the Bacteroidetes phylum have long-standing associations with animals. So, I believe this class of lipids may have unexplored relevance to animal biology that may have ancient roots.”
Reference: Alegado, Brown, Cao, Dermenjian, Zuzow, Fairclough, Clardy & King. 2012. Bacterial regulation of colony development in the closest living relatives of animals. eLife, citation to be confirmed.