Fighting bacteria with bacteria – common nose germ provides new weapon against superbugs
Our bodies are under siege, constantly fighting back assaults from disease-causing bacteria. But we are also home to many harmless bacterial species that are share our bodies to no ill effects. Now, it seems that these ‘commensals’ could be our hidden allies against their harmful cousins. In one such ally, a group of scientists has just discovered a potential new weapon against Staphylococcus aureus.
S.aureus is incredibly common, colonising the noses of a third of people in the USA, UK, Japan and other countries. Often, these colonies do nothing untoward, but if a full-blown infection sets in, the result can include life-threatening diseases like pneumonia, meningitis, toxic shock syndrome, endocarditis and sepsis. With the rise of MRSA and other staph strains that shrug off our most common antibiotics, the threat posed by this common nose bug has never been greater.
But S.aureus doesn’t have our noses to itself. It has to jostle for space with a close relative called Staphylococcus epidermidis. It’s the most common commensal in our noses and, indeed, the most common contaminating bacterium in laboratory equipment. S.epidermidis is harmless, except in people whose immune systems have been compromised. But more interestingly, it has the ability to stunt the growth of its more infamous cousin. Now, Tadayuki Iwase from Jikei University has isolated the protein it uses to do so.
Iwase swapped the noses of 88 volunteers and found that virtually all of them were colonised by S.epidermidis. However, S.aureus had only set up shop in just under a third. On the whole, the two bacteria seem to be able to co-exist in harmony, but Iwase found that some strains of S.epidermidis are anathemas to S.aureus.
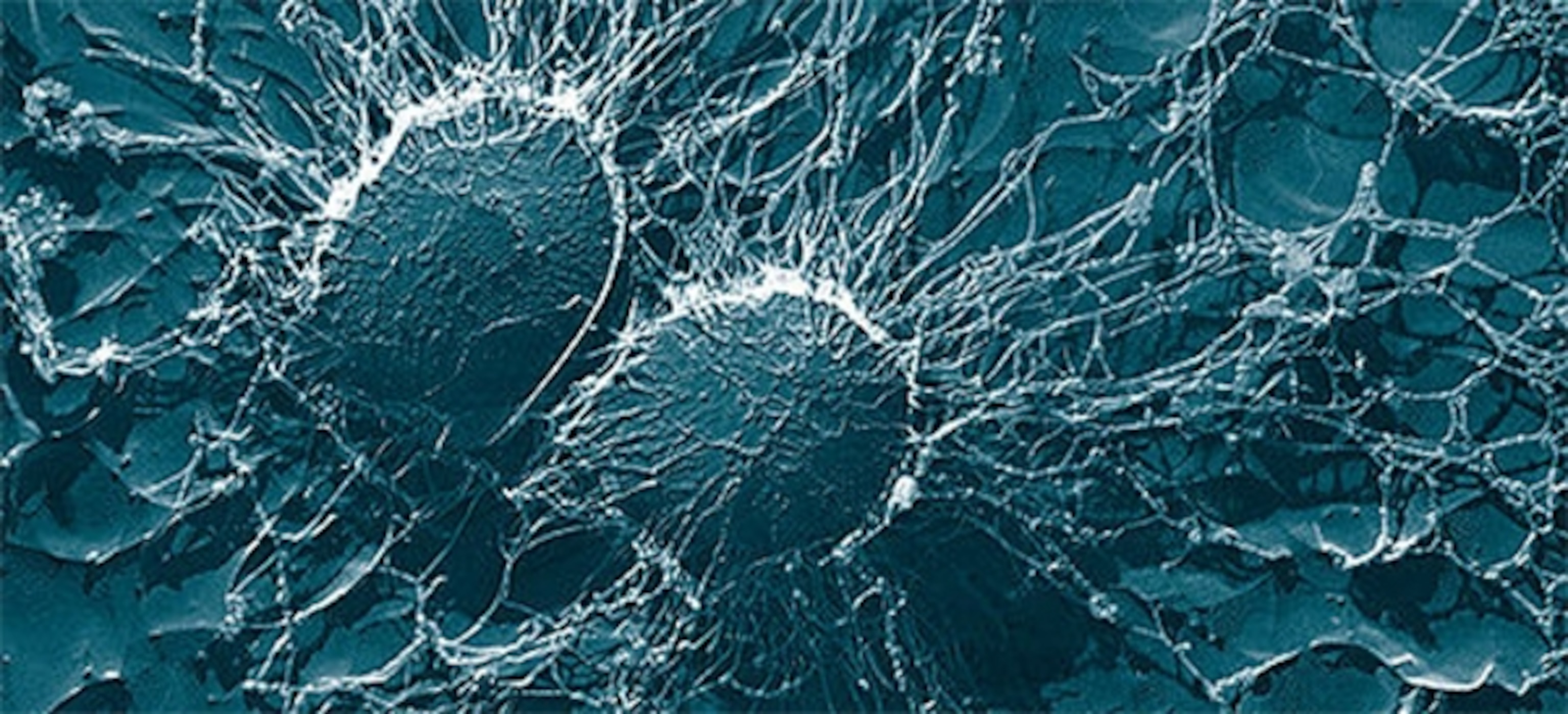
Specifically, they caused problems for S.aureus’s ability to set up biofilms, the bacterial equivalent of cities. Thousands of bacteria swarm within these communities, embedded in a slimy matrix of DNA, proteins and sugars. Within biofilms, bacteria are harder to kill, making them an important public health challenge. But according to Iwase, some strains of S.epidermidis not only prevent S.aureus from creating biofilms, they also destroy existing ones. People who were colonised by these defensive strains were around 70% less likely to be colonised by S.aureus.

To work out the weapon that was keeping the rival bacteria are bay, Iwase let cultures of S.epidermidis cut a swath through S.aureus biofilms and analysed their secretions when the destruction had reached its peak. He managed to isolate a single protein called Esp or ‘S.epidermidis serine protease’ in full. The protein was absent from strains that couldn’t wipe out S.aureus biofilms and present in strains that could. If Iwase gave the latter bacteria them a chemical that negates the Esp protein, or if he removed the esp gene from them entirely, they lost their competitive edge against S.aureus.
Esp even works in tandem with our own defensive proteins, including one called hBD2 (human beta-defensin 2) that’s secreted by our skin cells. Alone, hBD2 can kill bacteria but it’s a bit of a wimp about it, while Esp (for obvious reasons) has no bacteria-killing ability of its own. But together, their powers are far greater, and they effectively kill S.aureus, even when it was under the protection of biofilms. (The idea that the two proteins have co-evolved with one another is an intriguing question for another time.)
As a final test, Iwase introduced the competitive strains of S.epidermidis into the noses of volunteers who were already colonised by S.aureus. Sure enough, these transplanted bacteria eliminated their evolutionary cousins. Even a purified dose of Esp alone did the trick.
These experiments are very exciting. Humans are fighting a pitched (possibly losing) battle against staph and MRSA in particular, and our antibiotic arsenal is falling short. What better source of new weapons than other bacteria that have been fighting the same fight for millennia? Obviously, there’s a lot of work to do to turn Esp into a viable treatment, but this study is a promising first step.
Even better, it seems that, for some unclear reason, S.aureus can’t evolve resistance to Esp. With its biofilms under attack, you would expect S.aureus to quickly adapt, but after a year of culturing the two species together, Iwase couldn’t find any evidence that of resistance.
Reference: Nature http://dx.doi.org/10.1038/nature09074
More on Staphylococcus:
//