
How Leafcutter Ants Evolved From Farmers Into Cows
Humans have been farming crops for thousands of years, but we’re new arrivals to the agricultural scene. Leafcutter ants have been doing the same thing for millions of years.
If you like wildlife documentaries, you’ve probably seen leafcutters before, marching in vividly coloured columns with leaves held aloft in their jaws. These sliced fragments are carried back to the nest and fed to a fungus, which the ants then eat. Neither ant nor fungus can survive without the other. Their partnership has fuelled the leafcutters’ success—they come in more than 40 species, and have among the largest colonies of any ant.
Leafcutter ants are often compared to farmers, and the analogy works on many levels. They apply fertilisers to their crops, in the form of bacteria that provide nitrogen to the fungal gardens. They also use pesticides— they grow antibiotic-secreting bacteria that disinfect the fungi of parasites or competitors.

But you can also think of the ants are a dispersed cow, a single plant-digesting creature made of millions of bodies. The fungi are its guts.
When a cow eats grass, enzymes in its saliva start digesting the food before it reaches the animal’s stomach. Salivary enzymes also begin detoxify the poisons that plants use to deter hungry mouths.
The ants do something similar. When they first bring leaves to the nest, they deposit them at the top layer of the garden, where the fungus is at its thinnest, but the need for digestion is great. The ants supply this demand by eating the fungus at the garden’s lower depths, ingesting (but not digesting) its cocktail of enzymes. They carry these enzymes to the top of the garden and excrete them onto the fresh leaves in faecal droplets. These are the colony’s saliva. They ensure that the leaves start to decompose as soon as possible. And just like saliva, they make it easier for fungi further down the nest to access the rich supply of nutrients inside the leaves.

Scientists have known about this practice since the 1970s, but Jacobus Boomsma from the University of Copenhagen has spent the last few years working out its details. In 2010, he showed that the ants’ faeces contain six protein-busting enzymes, all of which come from the fungi. Most are manufactured in gongylidia—balloon-like clusters produced by the fungus, which the ants eat.
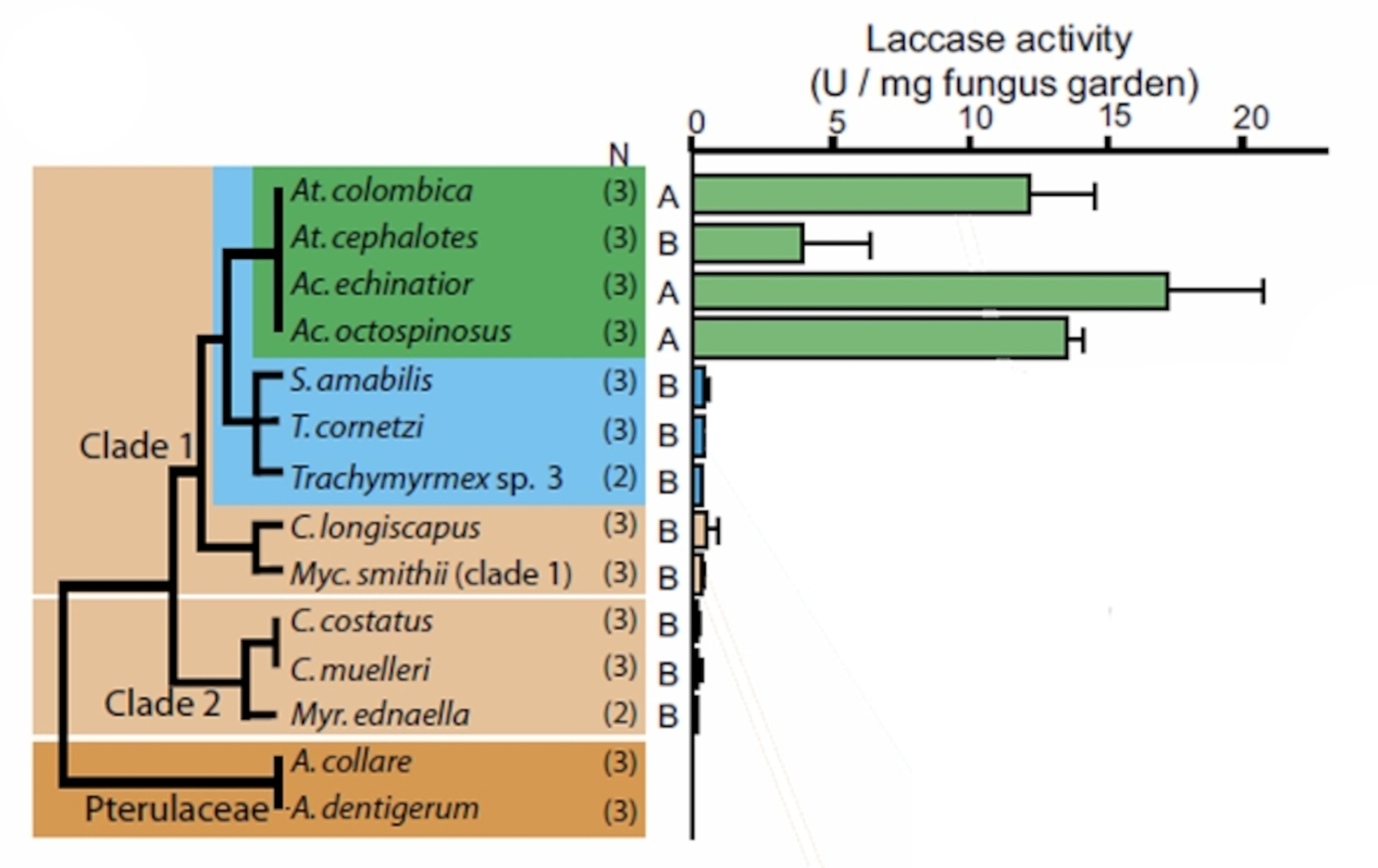
Now, Boomsma’s postdoc Henrik de Fine Licht has shown that the fungi also provide the ants with an enzyme called laccase, which breaks down poisons like tannins and flavonoids found in the leaves—again, much like a cow’s saliva.
De Fine Licht found that leaf-cutting ants have higher levels of laccase in their fungal gardens than other related species. The enzyme is also particularly abundant at the top of the garden, acting as a first response to the influx of poison. The workers expel laccase in their faecal droplets, but the enzyme clearly comes from the fungus—if the workers aren’t allowed to eat, their poo is laccase-free.
The fungus’ genome has nine laccase genes, but the enzyme carried by the ants is the work of just one of these—LgLcc1. This is also the only gene of the nine that is strongly activated in the gongylidia that the ants eat.
De Fine Licht showed that this particular gene has changed a lot since these fungi formed partnerships with ants, in ways that probably cemented the success of the leaf-cutters. Ants have been farming fungi for around 50 million years, but they originally fed their gardens with bits of dead plants, flower leaves and insect poo. This menu was relatively low in nutrients, but also low in toxins and easy to digest. The ants were more like farmers then, supplementing their diet of fungus with other sources of food.
But in the last 8 and 12 million years, some of the farmers evolved into leafcutters, feeding their gardens with freshly cut plant matter. And as they made the transition, their fungi evolved too. De Fine Licht compared LgLcc1 to similar genes from other fungal strains, cultivated by ants that farm fungus but don’t cut leaves. He found strong signatures of positive selection—where new mutations are embedded in a population because of the advantages they provide.
The team think that these mutations allowed the enzyme to survive a trip through an ant’s body, so that it could be carried to the top of the nest where the freshest and most heavily poisonous leaves are found. By neutralising the toxins at the point of entry, the ants and fungus jointly solved a digestion challenge.
This doesn’t require any planning on the part of the ants. They just went about their normal business of chewing up the fungus and defecating at a different part of the nest. If the fungus had the mutated form of laccase, its ant partners would do better than those whose fungi didn’t have these advantageous changes.
Something similar happened with human farmers. They unwittingly selected for genetic changes in crops like wheat and corn that make them easier to farm. As a result, domesticated wheat bears little relation to its wild ancestors. Its grains grow larger and stay on the stem, which makes them easier to harvest. In wild versions, the smaller grains break off and fall to the ground. Corn and maize have transformed to an even greater extent from their wild ancestor, teosinte.
The ants have done the same for their domesticated fungus, but they have gone one step further. Their partnership has become so intimate that, unlike us, they can no longer survive without their particular crop. Both ant and fungus have effectively become different parts of a single grazing entity.

Reference: De Fine Licht, Schiott, Rogowska-Wrzesinska, Nygaard, Roepstorff & Boomsma. 2012. Laccase detoxification mediates the nutritional alliance between leaf-cutting ants and fungus-garden symbionts. PNAS http://dx.doi.org/10.1073/pnas.1212709110