
The many yous in you – what Lydia Fairchild has in common with a sponge and an anemone
Lydia Fairchild was confused. She had applied for state benefits to look after her three children, but according to DNA tests, she was not their mother. It was ridiculous – she knew full well that the children were hers, but she was being taken to court nonetheless.
This happened in 2002, but Fairchild’s case has striking parallels with one that cropped up just this year, involving a Mediterranean sponge called Scopalina lophyropoda. French scientists Andrea Blanquer and Maria-J Uriz found that around a quarter of the sponge’s larvae are genetically distinct from the parents that they come from. Somehow, they had inherited genes from a different source.
Sponges are about as far away from humans as you could imagine an animal being – their bodies are just two layers of cells, curved and folded into tubes and chambers. But even though their bodies are worlds apart, the mysteries of both Lydia Fairchild and S.lophyropoda had the same answer.
Both of them are chimeras, living things that are formed when two or more fertilised eggs fuse together. The chimeras of myth were monsters that combined parts of lion, snake and goat. Real chimeras merely have two or more groups of cells with distinct sets of genes. You get two individuals for the price of one.
It looked like Fairchild and the sponge were not the parents of their own offspring because scientists had taken DNA sample from the wrong part of them. In Fairchild’s case, the DNA in her skin, hair and blood didn’t match those of her children, but DNA from a cervical smear eventually did. In the case of the sponge, DNA taken from other parts of the orange, blobby mass, proved to be a match for the mysterious larvae.
Chimeras challenge our notions of identity. We’re used to the idea that you get one individual per body. If you took a skin cell from your foot, a hair from your head, and a muscle cell from your heart, you’d find that they are all genetically identical. Every one of your cells came from a single predecessor so, barring a few copying errors, you carry the same genes everywhere. That’s patently not the case for chimeras. The DNA in two parts of their bodies can be so different that they might as well belong to two different individuals, which just happens to share the same shell.
On land, chimeras are fairly rare – Fairchild’s case is probably the exception rather than the rule. But there are species where chimerism is far more common. For example, the majority of marmosets are chimeras, carrying their own genes as well as their siblings. The upshot of this is that male marmosets regularly fertilise females with their brother’s genes.
But to really find hordes of chimeras, you need to look in the oceans. Colonial animals like corals and sea squirts regularly form fusion individuals, but two new studies have shown that sponges and sea anemones belong to the chimeric club too.
Blanquer and Uriz first discovered the chimeric nature of their sponge by looking at repetitive bits of DNA called microsatellites. These sequences vary in number from individual to individual, and they act as a genetic fingerprint. In humans, they can be used to identify individuals in forensic work or to establish family connections. In the sponge, they showed that some larvae had inherited genes that their parent didn’t have.
When Blanquer and Uriz snipped four pieces from 13 sponges, they found that these pieces had identical microsatellites in only three cases. For five of the sponges, all four pieces were genetically distinct! Despite looking like a uniform orange blob, some sponges were actually made up of several “individuals” or “genotypes”, with distinct but invisible boundaries between them. Each separate part could reproduce and form new sponges, but they all behaved as a single large individual, going through life together but apart.
This explains why S.lophyropoda is far more genetically diverse than expected. It lives in small, patchy areas and its larvae stay in the same place as their parents. You would expect its populations to be full of clones and inbreeding, but not so. In fact, they harbour a surprisingly diverse array of genes.

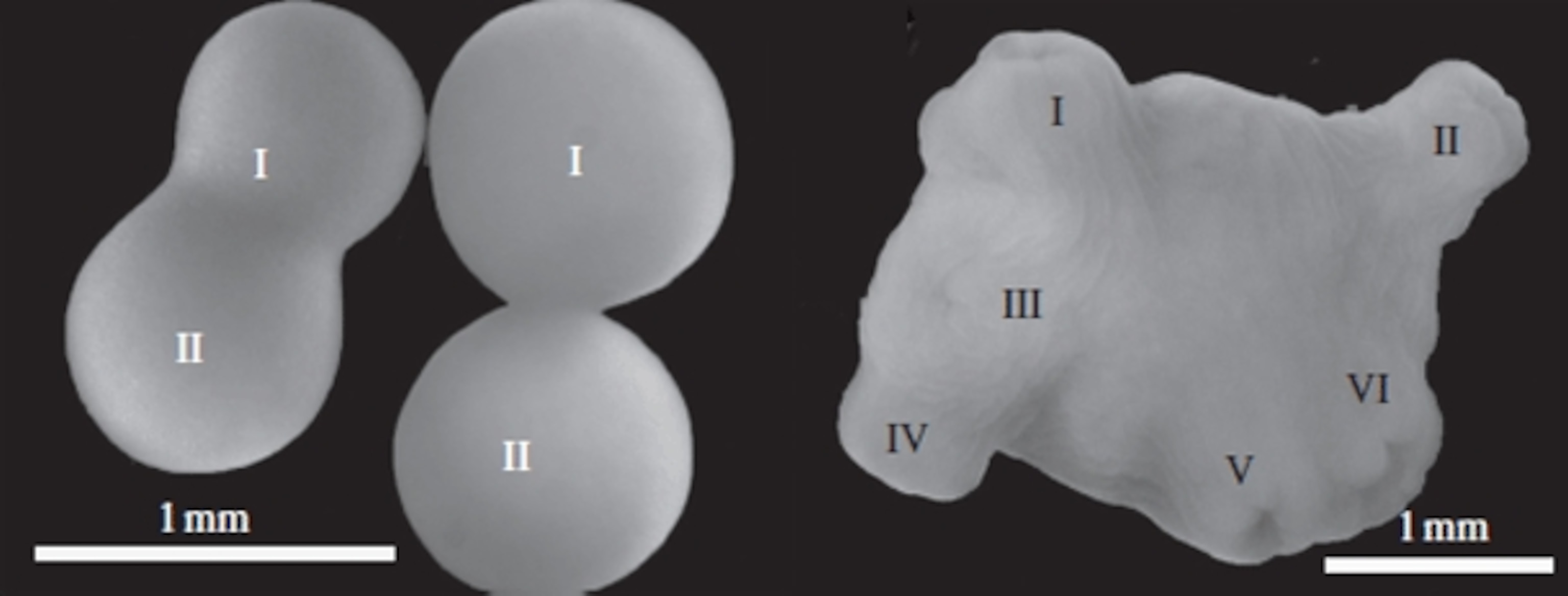
Annie Mercier from Memorial University in Canada also found that chimeras are surprisingly common among another simple sea animal – the dahlia anemone (Urticina feline), which captures prey with a ring of pretty tentacles. By eye, Mercier could see that at least one in every hundred anemone embryos is a chimera.
Most of these were fusions consisted of two embryos, but around a fifth were chaotic mergers between several individuals – six of them, in the picture below. In some cases, the embryos were joined by a thin thread of tissue, the anemone version of human Siamese twins. Others were far more extensively fused, and the eggs developed into two-‘headed’ adults that shared the same gut and nervous system. When Mercier prodded one, the other contracted.
This is just the tip of the iceberg. Mercier only picked out chimeras that were clearly mash-ups of multiple eggs. But she noticed that up to 90% of the anemone embryos were bigger than expected, given the size of the eggs. It’s possible that the default state for the anemone is not a single individual but a chimera, whose genetic fusion is so perfect that biologists can’t spot the seams. It’s only the more imperfect fusions that stand out.
Why form chimeras at all? For both the sponge and the anemone, chimeras are larger and they could grow faster and produce more young themselves. Alternatively, having two or more genomes could arm the animals with extra genetic diversity, useful for adapting to changing environments or resisting disease.
That’s certainly important for the sponge, which lives in seasonal waters with dramatically changing conditions. One genotype could grow particularly quickly in some conditions, but lose out in others. This could explain why many Mediterranean sponges seem to spend decades continuously shifting in shape and size, without actually growing any bigger. If the going gets tough, ‘you’ just expand the part of ‘you’ that’s best suited for the conditions at hand.
These discoveries raise some fascinating questions. How do these animals recognise their own bodies or their kin from intruders and strangers? Did the breakdown of this recognition lead to the evolution of social corals from solitary anemones? Do the different genotypes ever fight it out to the detriment of the entire ‘individual’? (Indeed, in sea squirts – a colonial animal that often forms chimeras – the partners sometimes live in harmony, but one sometimes kills and absorbs the other.)
And perhaps most importantly, have scientists underestimated the population sizes or diversity of chimeric animals, because they assumed that there’s only one individual per body? And how many chimeras are out there waiting to be discovered?
References: Blanquer, A., & Uriz, M. (2011). “Living together apart”: the hidden genetic diversity of sponge populations Molecular Biology and Evolution DOI: 10.1093/molbev/msr096
Mercier, A., Sun, Z., & Hamel, J. (2011). Internal brooding favours pre-metamorphic chimerism in a non-colonial cnidarian, the sea anemone Urticina felina Proceedings of the Royal Society B: Biological Sciences DOI: 10.1098/rspb.2011.0605
Images: anemone by Asbjorn Hansen