Nocturnal mammals see in dark by turning displaced DNA into lenses
Nocturnal animals face an obvious challenge: collecting enough light to see clearly in the dark. We know about many of their tricks. They have bigger eyes and wider pupils. They have a reflective layer behind their retina called the tapetum, which reflects any light that passes through back onto it. Their retinas are loaded with rod cells, which are more light-sensitive than the cone cells that allow for colour vision.

But they also have another, far less obvious adaptation – their rod cells pack their DNA in a special way that turns the nucleus of each cell into a light-collecting lens. Their unconventional distribution is shared by the rods of nocturnal mammals from mice to cats. But it’s completely opposite to the usual genome packaging in the rods of day-living animals like primates, pigs and squirrels, and indeed, in almost all other eukaryotic cells.
In our cells, massive lengths of DNA are packaged into small spaces by wrapping them around proteins. These DNA-protein unions are known as chromatin, and they come in two different forms. Euchromatin is lightly packed and resembles a string of beads. Wrapping DNA in this way puts it within easy reach of other proteins and allows its genes to be actively transcribed. But imagine scrunching up that string of beads and you get heterochromatin – a tight, condensed ball of repressed genes that proteins cannot reach.
The two forms of chromatin are found in different areas, with euchromatin spread throughout the nucleus and heterochromatin concentrated at its edges. That pattern is nigh-universal and it applies from amoebae to plants to animals. There are only a few exceptions to this rule, including a minority of single-celled species and surprisingly, the rod cells in the eyes of nocturnal mammals. Now, Irina Solovei from the Ludwig-Maximilians University in Munich had found that this inverted distribution helps these species to see in the dark.
Solovei began by looking at the eyes of mice, which are almost entirely comprised of rod cells. She used fluorescent probes to reveal the location of the two forms of chromatin. In most eye cells, she saw the usual pattern in cross-section – a centre of euchromatin surrounded by a heterochromatin ring. The rod cells, however, had the opposite arrangement, with active euchromatin around a core of silent heterochromatin.
The mice aren’t born with this topsy-turvy arrangement in their rods – rather, there’s a switchover that starts on its sixth day of life and takes a couple of weeks to complete. During this relocation, the cell’s nucleus changes shape from an ellipse to a sphere, and shrinks by about 40%.
The inverted pattern wasn’t just a feature of mouse eyes – Solovei found the same thing in the rods of other animals with nocturnal habits. She looked at the eyes of 38 species hailing from 10 different orders, and found a clear link between the DNA in their rods and their daily lifestyles.
Species that are active at night, like cats, rats or opposums, have the inverted model. Day-lovers like horses and pigs have the conventional one. Deer, rabbits and “crepuscular” creatures that are active at dawn or dusk showed both patterns or, in the case of cows, an intermediate one. These relationships even held between members of the same group – among the primates, for example, nocturnal mouse lemurs have the inverted pattern, while diurnal macaques have the standard one.
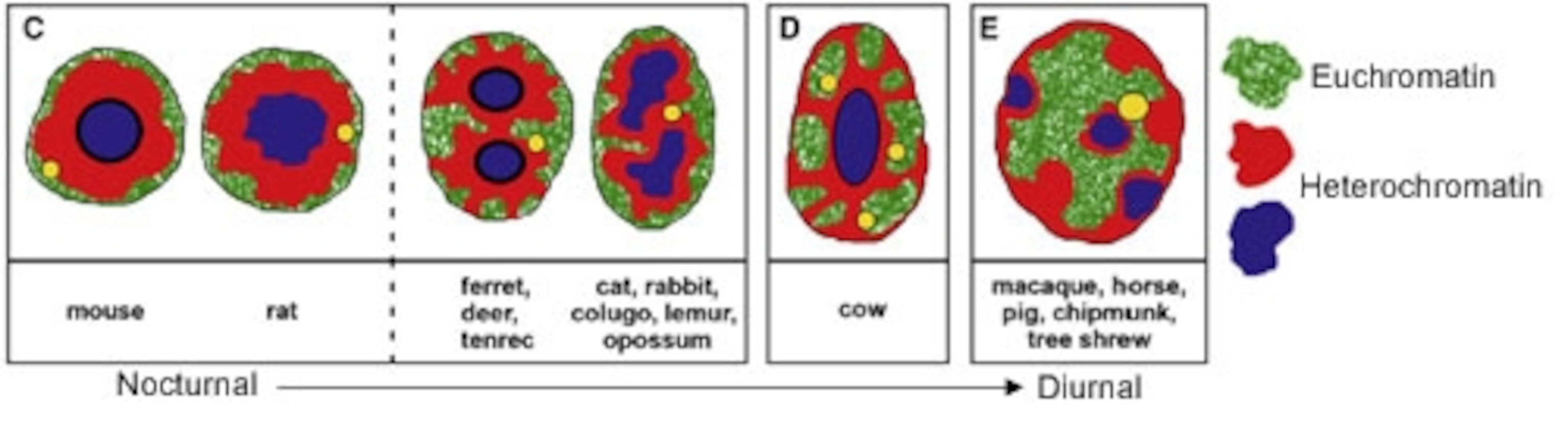
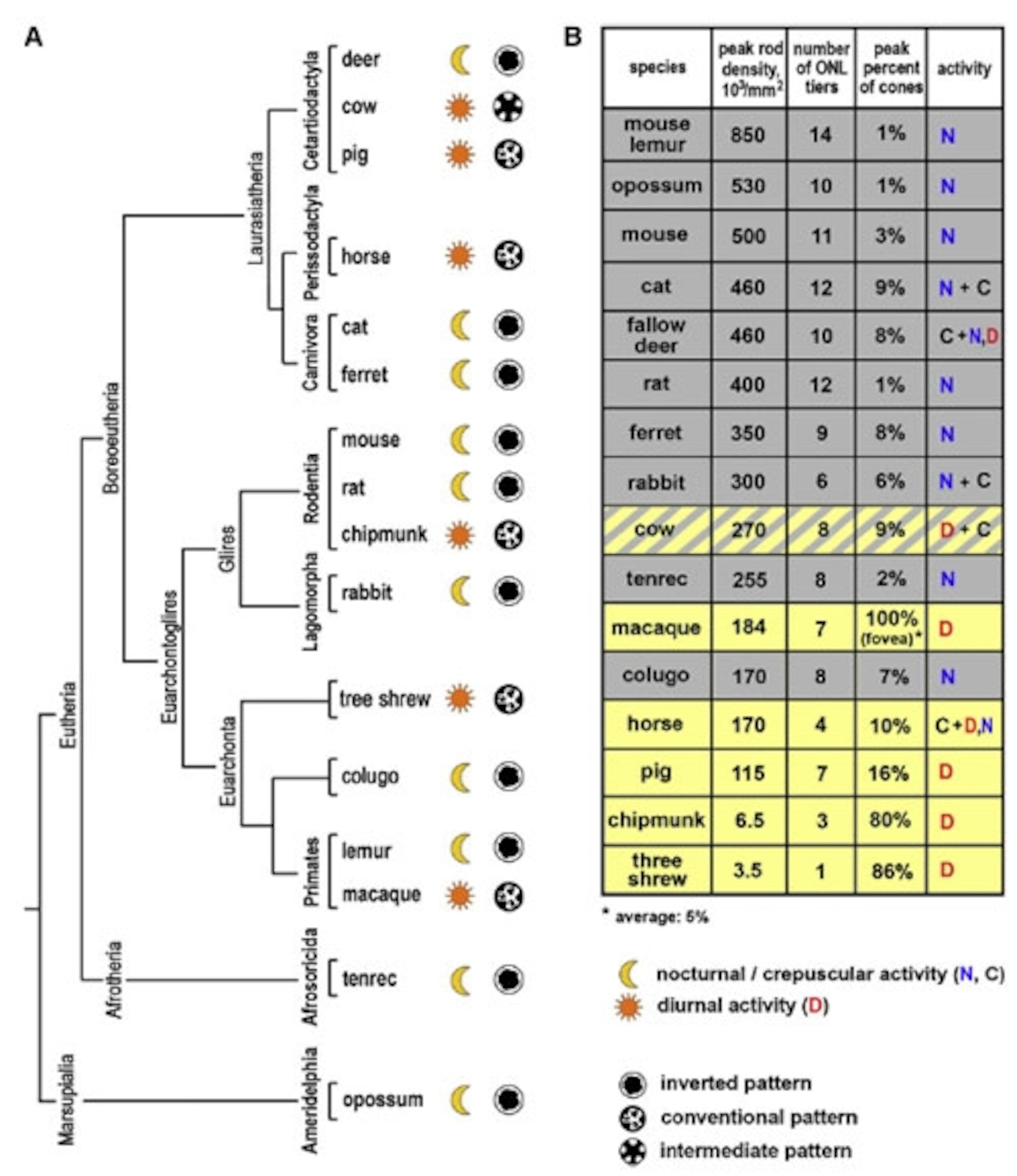
To Solovei, this clear-cut connection was an obvious sign that the nocturnal arrangement of chromatin helps mammals to see in the dark. By running computer simulations and studying single rods under the microscope, she discovered that the rearranged DNA transform the nuclei into small lenses, collecting and funnelling light through the retina.
This light collection is massively important, when you consider that mammalian eyes have a back-to-front, incompetent design. The retina’s light-sensitive rods and cones are right at the back, hidden behind a tangled weave of nerves and blood vessels. Even if light manages to get through, the rods themselves are arranged awkwardly, so that the segments that actually contain the light-sensitive pigments are blocked by columns of DNA-rich nuclei. The eyes of nocturnal mammals have so many rods that they present a thick wall of nuclei in front of the light-sensitive segments.
That would be a problem were it not for the rearranged chromatin. In its normal configuration, it would just scatter any light that found its way to the retina. But push the euchromatin to the edges, and suddenly the nuclei start passing light down from one to another. The structure ensures that as many photons as possible reach their final destination, allowing nocturnal mammals them to see at light levels a million times lower than those that are available to us during the day.
Based on the fossil record and molecular evidence, Solovei thinks that this inverted chromatin pattern evolved once in the early stages of mammal evolution, when our ancestors were still scurrying, creatures of the night. Species that once again ventured out in daylight reacquired the conventional pattern independently.
That may seem unlikely, but remember that this pattern is standard in virtually all eukaryotic cells. Such constancy implies that the normal arrangement has some important advantages to it. Presumably, it plays an important role in controlling which genes are switched on or off and indeed, we know that changing a gene’s geography – where it’s located in the nucleus – can be used to regulate it. Disrupting this system must have come at a price, and one that was only outweighed in nocturnal mammals by the vital need to see in the dark.
Reference: Solovei, I., Kreysing, M., Lanctôt, C., Kösem, S., Peichl, L., Cremer, T., Guck, J., & Joffe, B. (2009). Nuclear Architecture of Rod Photoreceptor Cells Adapts to Vision in Mammalian Evolution Cell, 137 (2), 356-368 DOI: 10.1016/j.cell.2009.01.052
Images: Cat by Vladimir Menko, others from Cell.