Here’s something you probably learned once in a biology class, more or less. There’s this molecule called DNA. It contains a long code that created you and is unique to you. And faithful copies of the code live inside the nucleus of every one of the trillions of cells in your body.
In a later class you may have learned a few exceptions to that “faithful copies” bit. Sometimes, especially during development, when cells are dividing into more cells, a mutation pops up in the DNA of a daughter cell. This makes the daughter cell and all of its progeny genetically distinct. The phenomenon is called ‘somatic mosaicism’, and it tends to happen in sperm cells, egg cells, immune cells, and cancer cells. But it’s pretty infrequent and, for most healthy people, inconsequential.
That’s what the textbooks say, anyway, and it’s also a common assumption in medical research. For instance, genetic studies of living people almost always collect DNA from blood draws or cheek swabs, even if investigating the tangled roots of, say, heart disease or diabetes or autism. The assumption is that whatever genetic blips show up in blood or saliva will recapitulate what’s in the (far less accessible) cells of the heart, pancreas, or brain.
Two recent reports suggest that somatic mosaicism is far more common than anybody ever realized — and that might be a good thing.
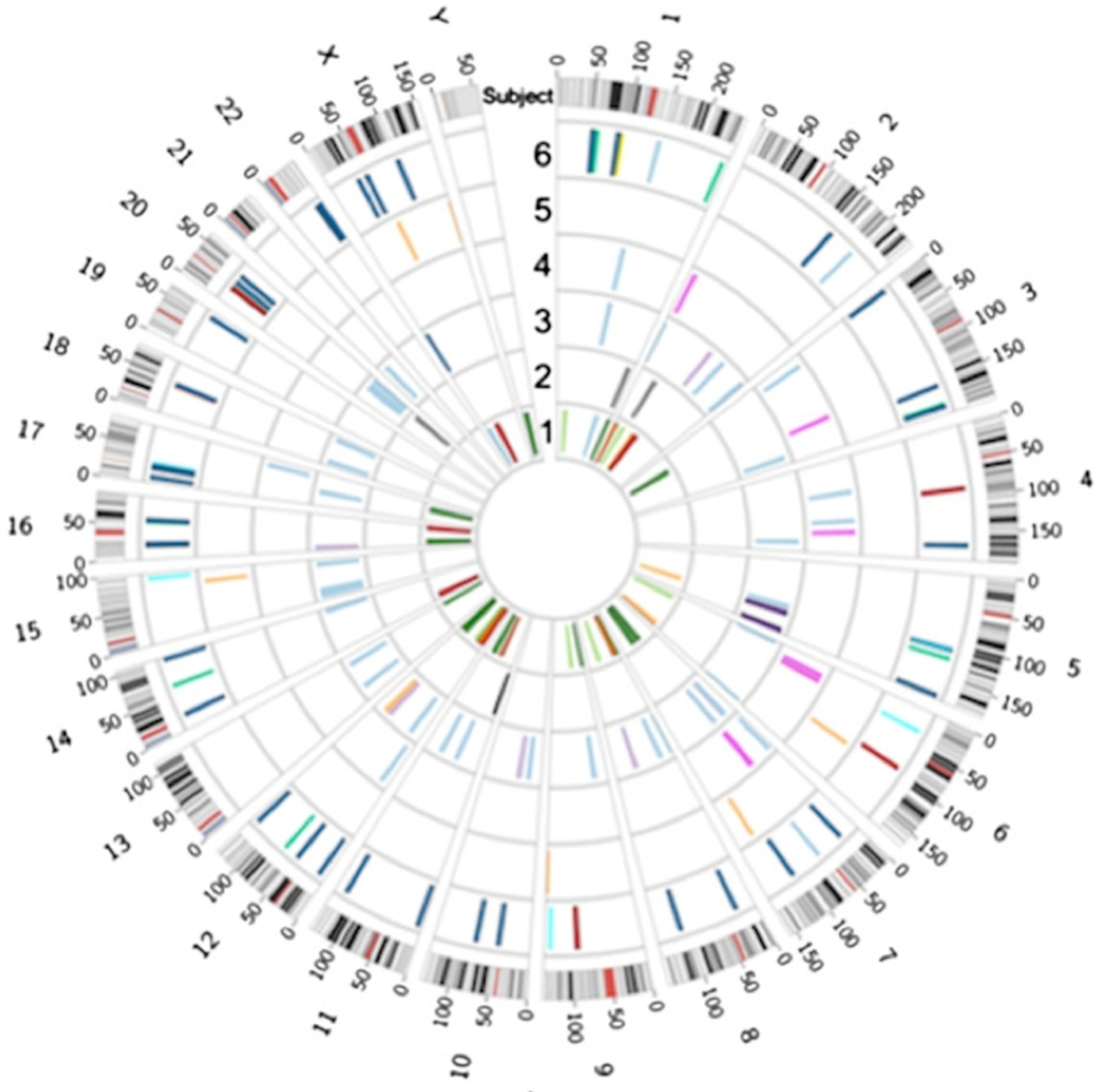
In the first study Michael Snyder and colleagues looked at cells in 11 different organs and tissues obtained from routine autopsies of six unrelated people who had not died of cancer or any hereditary disease.
Then the scientists screened each tissue for small deletions or duplications of DNA, called copy number variations, or CNVs. These are fairly common in all of us.
In order to do genetic screens, researchers have to mash up a bunch of cells and pull DNA out of the aggregate. That makes research on somatic mutations tricky, because you can’t tell how some cells in the tissue might be different from others. The researchers got around that problem by doing side-by-side comparisons of the tissues from each person. If one tissue has a CNV and the other one doesn’t, they reasoned, then it must be a somatic glitch.
As they reported in October in the Proceedings of the National Academy of Sciences, Snyder’s team found a total of 73 somatic CNVs in the six people, cropping up in tissues all over the body, including the brain, liver, pancreas and small intestine. “Your genome is not static — it does change through development,” says Snyder, chair of the genetics department at Stanford. “People knew that, but it had never been systematically studied.”
OK, but do somatic mutations do anything? It’s hard to tell, particularly because postmortem studies offer no living person to observe. Still, the scientists showed that 79 percent of the somatic mutations fell inside of genes, and most of those genes play a role in the cell’s everyday regulatory processes, like metabolism, phosphorylation, and turning genes on. So the somatic mutations could very well have had an impact.
In the last paragraph of their paper the researchers mention that the findings could also have big implications for studies of induced pluripotent stem (iPS) cells. This line of research is getting increasingly popular, for good reason. With iPS technology, researchers start with a small piece of skin (or…) from a living person. They then expose those skin cells to a certain chemical concoction that reprograms them back into a primordial state. Once the stem cells are created, researchers can put them in yet another chemical soup that coaxes them to differentiate into whatever type of cell the scientists want to study. You can see why it’s cool: The technique allows scientists to create cells — each holding an individual’s unique DNA code, remember — in a Petri dish. Researchers can study neurons of children with autism, for example, without ever touching their brains.
Trouble is, several groups have reported that iPS cells carry mutations that the original skin cells don’t have. This suggests that something screwy is happening during the reprogramming process, defeating the whole purpose of making the cells. (Fellow Phenomena contributor Ed Yong wrote a fantastic post about the hoopla last year.)

But that last paragraph of Snyder’s study offers a bit of hope. What if the mutations that crop up in iPS cells actually were in the skin cells they came from, but just didn’t get picked up because those skin cells were mixed with other skin cells that didn’t have the mutations? In other words, what if skin cells, like all those other tissues they looked at in the paper, are mosaics?
The second new study, published last month in Nature, finds exactly that.
Flora Vaccarino‘s team at Yale sequenced the entire genome of 21 iPS cell lines, three each from seven people, as well as the skin cells that the iPS cells originated from. It turns out that each iPS line has an average of two CNVs and that at least half of these come from somatic mutations in the skin cells. (The researchers used special techniques for amplifying the DNA of the skin cells, so that they could detect CNVs that are present only in a fraction of the cells.)
That means two things. First, researchers using iPS cells can exhale. Their freaky reprogramming process doesn’t seem to create too much genetic havoc in the iPS cells. And second, somatic mosaicism happens a lot. Vaccarino’s study estimates that a full 30 percent of the skin cells carry somatic mutations.
Our widespread mosaicism may have implications for certain diseases. Somatic mutations have been strongly linked to tumors, for example, so it could be that people who have a lot of mosaicism are at a higher risk of cancer. But there’s also a positive way to spin it. Somatic mutations give our genomes an extra layer of flexibility, in a sense, that can come in handy. Snyder gives a good example in his study. If you have a group of cells that are constantly exposed to viruses, say, then it might be beneficial to have a somatic mutation pop up that damages receptors on the cell that viruses can latch on to.
But there’s likely a more parsimonious explanation for all of those genetic copying mistakes. “When you’re replicating DNA, there’s a certain expense to keep everything perfect,” Snyder says, meaning that it would cost the cell a lot of energy to ensure that every new cell was identical to the last. And in the end, he adds, that extra expense may not be worth it. “Having imperfections could just be an economically beneficial way for organisms to do things.”
*
Photos from Shannon O’Hara and James Diin, courtesy of National Geographic’s My Shot
