Gut Microbes Contribute to Mysterious Malnutrition
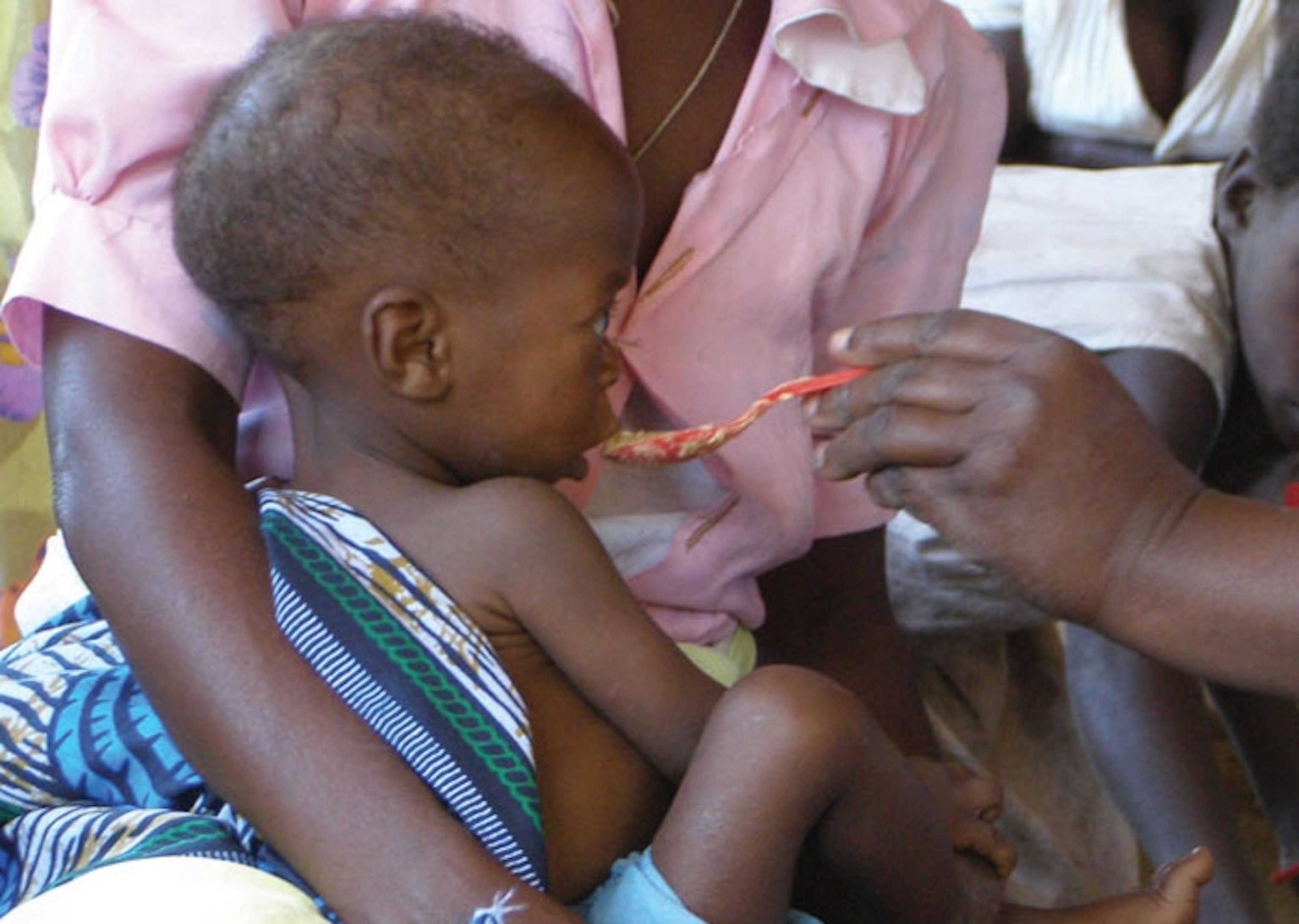
In 2007, Indi Trehan was approached by a Malawian woman whose two children were severely malnourished. Trehan, a paediatrician from Washington University in St Louis, had seen many such cases. Malawi has one of the highest rates of child mortality in the world, and half of these deaths are due to malnourishment. Trehan had come to the country to help.
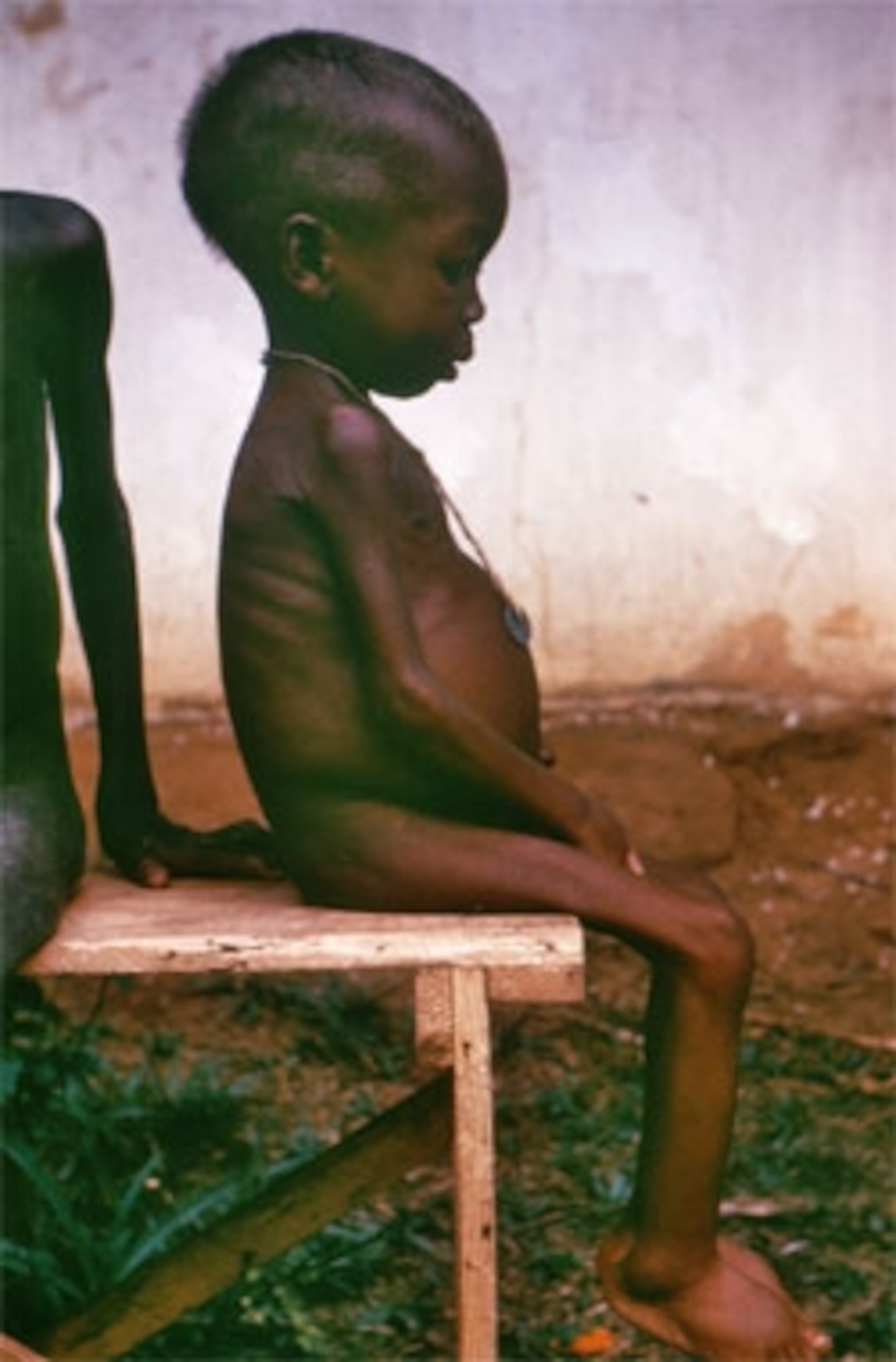
But these children surprised him. They were identical twins. One had a type of malnutrition called marasmus, and looked emaciated and skeletal. The other had kwashiorkor, a very different condition where fluids leak from blood vessels and don’t get reabsorbed. These children have puffy swollen limbs, distended stomachs and irritable moods. Their skin starts to break down and often becomes infected.
The two twins were genetically identical, had been raised in the same environment, and had both eaten the same poor diets. But they had ended up with incredibly different conditions. Why? Trehan was stumped and he emailed his colleague Mark Manary, who had also come from the US to Malawi. Manary had seen the same patterns, and had a hunch about what was causing them. He wrote back: “Yeah, I’ve been talking to this Jeff Gordon guy.”
Jeff Gordon works at the same university as Trehan and Manary, but not in child health. He’s an expert on the microbiota—the trillions of microbes that live in our bodies. These partners act like one of our organs, providing us with nutrients, safeguarding our health, and helping us digest our food. In 2006, Gordon showed that the microbiota can affect our risk of becoming obese. Fatter people have guts that are teeming with Firmicutes bacteria but low in the Bacteroidetes group. When Gordon transplanted the gut microbes from fat mice into lean ones, the recipients started extracting more energy from their food and put on weight.
If these bacteria can influence obesity, could they also be involved in its polar opposite—malnutrition? At first, Trehan through it was unlikely. Even other microbiota scientists were sceptical. “Jeff has been very excited about this idea for years, but I thought it was one of the less plausible connections between the microbiota and human disease,” says Rob Knight, one of Gordon’s frequent collaborators. “But the potential significance was so great that it was clearly worth finding out if it were true.”
It was. Gordon, Knight, Manary, and Trehan joined forces and found clear evidence that the microbiota of Malawian twins are sometimes involved in kwashiorkor. Even between identical twins, there were stark differences between the gut microbes of children with and without the condition. And when the team transplanted the microbes of affected children into mice, the rodents lost weight.
“We’re hesitant to put this all down to the microbiome but it’s the first real player that has been proven,” says Trehan. “Is the microbiome change coming first and leading the kwashiorkor, or responding to something else? It’s a chicken and egg problem, but at least we’re in the right section of the grocery store!”

Twins and transplants
Kwashiorkor has long been shrouded in mystery. Why is it so common in southern Africa, and not in other parts of the world where malnourishment is common? And what actually causes it? No one really knows. The most common explanation (featured prominently on Wikipedia) is that it’s the result of a protein-poor diet. But that can’t be the whole story. Kwashiorkor children don’t eat any less protein than those with marasmus, and some of them recover despite eating little protein, while others fail to improve on protein-rich food.
Other scientists have pointed their fingers at environmental toxins, but that can’t account for the condition’s bizarre pattern. “You have children living in the same village, eating the same food, having the same exposure to infections, and facing the same cycle of food insecurity,” says Trehan. “From the outside, these kids look the same”, but only some of them get kwashiorkor. Even identical twins frequently become discordant for the condition—that is, one gets it and the other doesn’t.
Twins might add to kwashiorkor’s mystery, but they’re also helping to dispel it. They share both genes and environment, allowing the team from Washington to rule out these other factors (or at least, to reduce their impact). “The twins provide you control when you don’t have it,” says Trehan.
The team recruited 317 pairs of twins and regularly collected their stool samples until they reached the age of three. They eventually focused on 24 pairs of same-gender twins—9 of them stayed healthy, while in the other 13, one child developed kwashiorkor and the other did not.
As the healthy, well-nourished children got older, their gut bacteria started wielding an increasingly diverse repertoire of genes. But the bacteria of kwashiorkor children stagnated. Their arsenal of genetic tools never changed.
The team fed all the starving children with “ready-to-use therapeutic food” (RUTF)—a fortified blend of peanut paste, sugar, vegetable oil and milk, which has become a standard treatment for malnutrition. The paste had a brief effect on the children’s bacteria, but as soon as they reverted to their normal Malawian diet, their microbes also bounced back to their earlier impoverished state.
Next, the team took the gut bacteria from three pairs of twins and transplanted them into germ-free mice, which lack their own gut microbes. In two of these three cases, the animals that received bacteria from the kwashiorkor twin lost more weight than those that were loaded with the healthy twin’s bacteria. So, a child with kwashiorkor can effectively transmit their symptoms to a mouse by donating their gut microbes.
“For a first demonstration, we would not expect it to work every time,” says Knight, who also would have liked to have transplanted the microbes of more than three twin pairs. However, “these experiments are cutting-edge, and limited by cost and facilities,” he says. For now, it seems that the microbiome is an important player in at least some cases of kwashiorkor.
A vicious cycle
The microbes aren’t the whole story. After all, the team didn’t find a consistent set of bacteria that’s associated with kwashiorkor. They also found that the kwashiorkor bacteria only affected mice that were fed with the equivalent of a Malawian diet—a nutrient-poor menu of corn flour and vegetables. If the mice ate a standard rodent chow, they didn’t lose much weight no matter whose bacteria they were carrying.
So diet’s important, but that’s not the whole story either. If it were that simple, the RUTFs would have shifted the microbiomes of kwarshiorkor children into the mature communities of their peers. They didn’t. Kwashiorkor may be a type of malnutrition, you won’t always solve it just by feeding kids with nutritious food.
The team found a few clues as to how the Kwashiorkor microbes and the Malawian diet work together. For example, this combo could conspire to starve a child’s body of sulphur. This crucial element plays an important role in many of our enzymes, and we mostly get it from our diet. Sulphur is part of amino acids like cysteine and methionine, which are more common in animal and cereal proteins. The Malawian diet contains very little of either, so is naturally low in sulphur. What little there is gets consumed by a bacterium called Biophilia wadsworthia, which is more common in the guts of children with kwashiorkor. This microbe could worsen the effects of the already-deficient diet.
Here’s another connection: The urine of the kwashiorkor mice contained lower levels of chemicals that are produced in the citric acid cycle—a chain reaction that our cells use to produce energy. The team found evidence that the kwashiorkor microbes are interfering with the cycle, making it harder for the children to harvest energy from their food—food that contains very little energy to begin with.
In an accompanying editorial, David Relman from Stanford University suggests a more complicated scenario, which he calls a “vicious cycle of undernutrition”. A poor diet can impair a child’s immune system, changing its ability to control its gut passengers and opening the door to harmful infections, which change the microbiome even further. These disrupted communities play havoc with the gut’s ability to absorb nutrients, which leads to even worse malnutrition, immune problems, and so on.
Relman sees the gut as an ecosystem, much like a lake or a forest. It doesn’t change in simple ways. It can shift into a stable and unhealthy state, that’s very difficult to get out of. To do so, you probably need several angles of attack. A better diet might be one. Changing the microbiome could be another. Perhaps we could tailor diets to nourish not just children, but their bacteria. Maybe we could provide molecules that improve the communication between the child’s cells and their microbe partners. And maybe these approaches will need to be tailored to each individual.
Breaking the cycle
For now, we don’t know enough to do any of this. “[Our study] is very far removed from practical implications,” says Trehan. “If you tracked a microbiome, you could predict whether a child has kwashiorkor, but the tech is slow and not ready for clinical use.”
But the technology is getting faster and cheaper. “The experience of cell phones and mobile Internet have shown that new technologies with clear and obvious benefits can move fast even into the developing world,” says Knight. He suggests that doctors may one day be able to transplant a sick child’s microbiome into a germ-free mouse, work out the foods they need to treat the mouse, and then try the same diet with the patient.
Eventually, we’d be able to skip the mouse altogether—a computer would analyse the microbes in a stool sample and print out an optimal diet. “This work is an important step towards both goals, although as always much remains to be done,” he says.
Reference: Smith, Yatsunenko, Manary, Trehan, Mkaksoya, Cheng, Kau, Rich, Concannon, Mychaleckyj, Liu, Houpt, Li, Holmes, Nicholson, Knights, Ursell, Knight & Gordon. 2013. Gut Microbiomes of Malawian Twin Pairs Discordant for Kwashiorkor. Science http://dx.doi.org/10.1126/science.1229000