If you don’t have an immune system, you don’t last long in this parasite-riddled world. Your body receives a steady stream of invaders–viruses, bacteria, and other pathogens–which it has to recognize and fight. In many cases, it’s a brutal battle with an ultimate goal of eradication. In other cases, the immune system simply keeps strangers in check, preventing them from spreading. As many as a third of all humans have cysts in their brains containing a single-celled parasite called Toxoplasma. As long as the parasite stays in its cyst, the immune system lets it be. If Toxoplasma breaks out and starts to multiply, however, the immune system picks off the new cells. And if people lose their immune system–due to HIV infection, for example–Toxoplasma runs rampant and causes devastating brain damage.
The cells and molecules we use to recognize these invaders are unquestionably amazing. What’s perhaps most amazing is that the immune system can learn. When a new pathogen turns up, our immune cells undergo a kind of interior version of natural selection. Over the course of several cell divisions, new variants emerge that do a better and better job of recognizing the newcomer. Our bodies can then mount a powerful, focused attack on, say, a particular strain of the flu. And once the immune system learns how to recognize that new enemy, it can store that memory away, enabling it to attack the same pathogen years later.
This is the sort of thing that people often have in mind when they refer to us as a “higher” form of life, and bacteria and viruses as a “lower” form. Bacteria are just simple individual cells. They’re not multicellular organisms that can dedicate billions of cells to making antibodies, spewing poisons, and carrying out the many other tasks required for an immune system to work. Viruses–forget about it–they’re just protein shells that package a few genes, which they insert into a host cell.
But the higher/lower dichotomy is a blinkered way to look at life. If you can’t believe that bacteria can have an immune system, then you will miss the clues that they, in fact, have one. And the evidence is overwhelming.
And the evidence is overwhelming.
Bacteria, after all, live in the same parasite-riddled world as we do. They may not get infected by the same pathogens that infect us, but they are continually hounded by viruses. A microbe that can defend itself against a virus will have a huge edge in the evolutionary race against its fellow microbes.
The threat of viruses has driven the evolution of some pretty impressive defenses. Bacteria make enzymes that lock onto certain, short sequences of DNA and slice them apart. When a virus injects its genes, these so-called restriction enzymes shred them into genetic confetti, so that they can’t take over the cell.
Our own immune system always runs the risk of turning against us and causing autoimmune disorders like arthritis and lupus. We have lots of safeguards in place to minimize that risk. Likewise, restriction enzymes are a dangerous defense, because they can chop up the distinctive stretches of DNA in a bacterium’s own genes. It avoids attacking itself by capping those sequences in its own DNA, so that the restriction enzymes can’t reach them.
The restriction enzyme defense is just one wing of the immune system in bacteria. Some species can muck up the production of new viruses, stealing their proteins before they can form shells. Others commit suicide upon infection, so as to avoid becoming an incubator for new viruses that would then kill their nearby relatives.
But the most impressive–dare I say it, most human–part of the bacterial immune system is its ability to learn. About forty percent of bacteria carry a set of genes known as CRISPR. When a virus invades these bacteria, they capture fragments of its DNA and insert them into their CRISPR genes. The bacteria then use those captured fragments as a guide for building weapons against the virus.
Here’s how this weaponizing works. In order to turn a virus’s genes into new virus proteins, a microbe must first make a copy of the gene in a molecule called RNA. CRISPR genes can produce RNA molecules with a matching sequence. They grab onto the virus’s RNA and prevent them from being turned into proteins. The virus factory grinds to a halt.
This defense helps bacteria withstand a virus infection, but it does more. The bacteria hold onto an invading virus’s DNA, so that they are now prepared for a fresh attack. And over time, bacteria can build up little libraries of these virus barcodes. A single bacterium may carry dozens of these viral barcodes. Last year, scientists at Indiana University surveyed the bacteria in people’s mouths and discovered 8,000 different viral barcodes–many of them corresponding to viruses scientists have yet to discover.
A single microbe can thus build up memories of its pathogens, in a manner reminiscent of the way we build up memories in our own immune system. But if you build up a healthy store of antibodies to various strains of flu, smallpox, and other diseases, all that knowledge dies with you. If you have children, they have to learn the same lessons all over again.
Not so for bacteria. When a microbe reproduces, it passes down its CRISPR genes and all of their viral barcodes to its descendants–including the ones it acquired in its own lifetime. Maybe Lamarck would have been better off as a microbiologist.
Now let’s swing around and consider the immune system from the pathogen’s point of view. If you can evolve a way to avoid the defenses of the immune system of your host, you will thrive where other pathogens are killed. This evolutionary pressure has led to all sorts of remarkable evasions carried out by the pathogens that make us sick. They camouflage themselves with human-like proteins; they attack key molecules, bringing our defenses to a standstill.
Viruses that infect bacteria have evolved their own set of tricks to evade the bacterial immune system. Last fall, for example, University of Cambridge scientists discovered viruses that carry an antidote for the suicide toxin made by their hosts. When the bacteria want to die, the virus forces them to live on. And just last month, University of Toronto scientists even discovered anti-CRISPR genes in viruses, which the viruses use to shut down the production of virus-killing molecules.
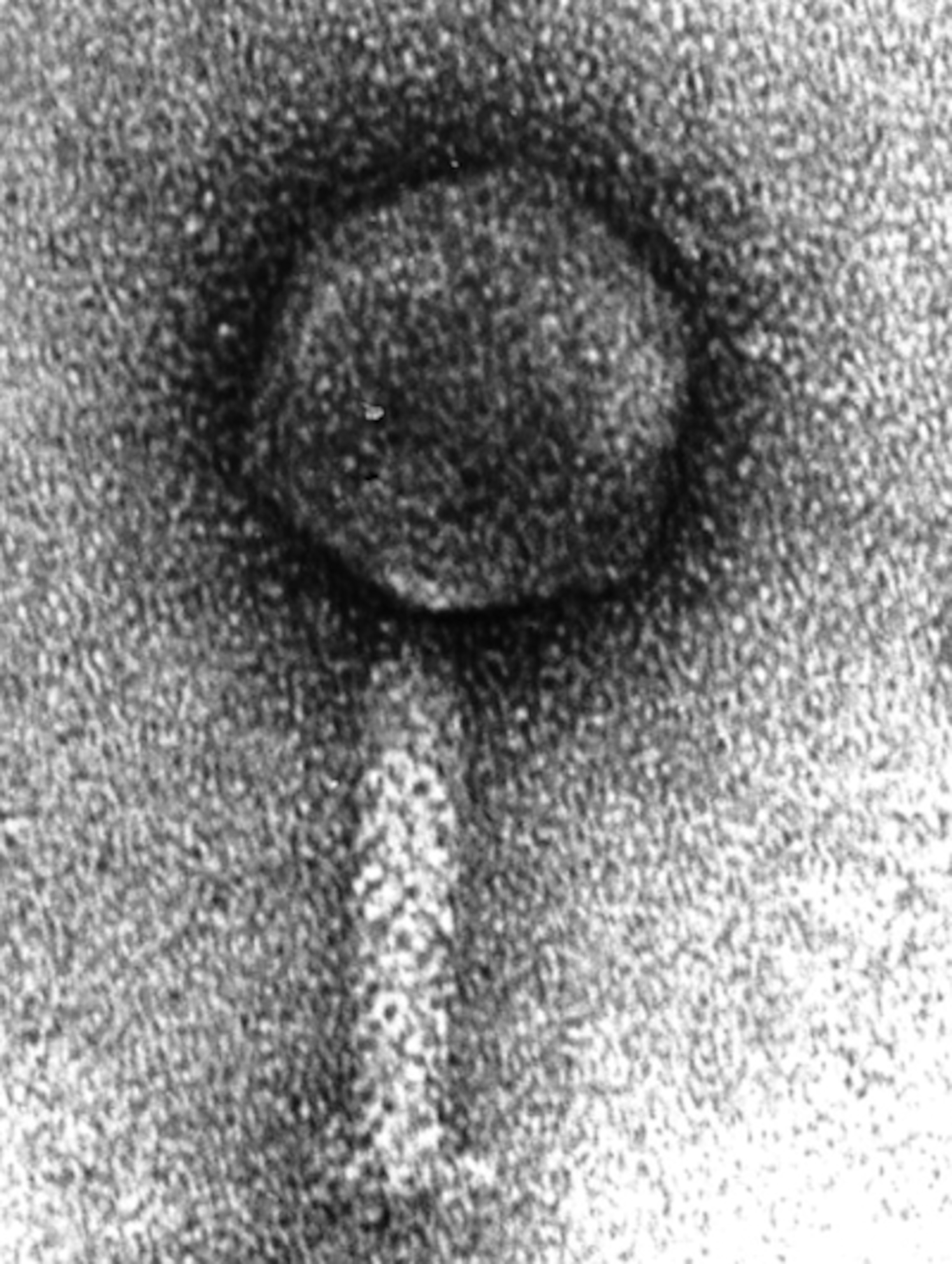
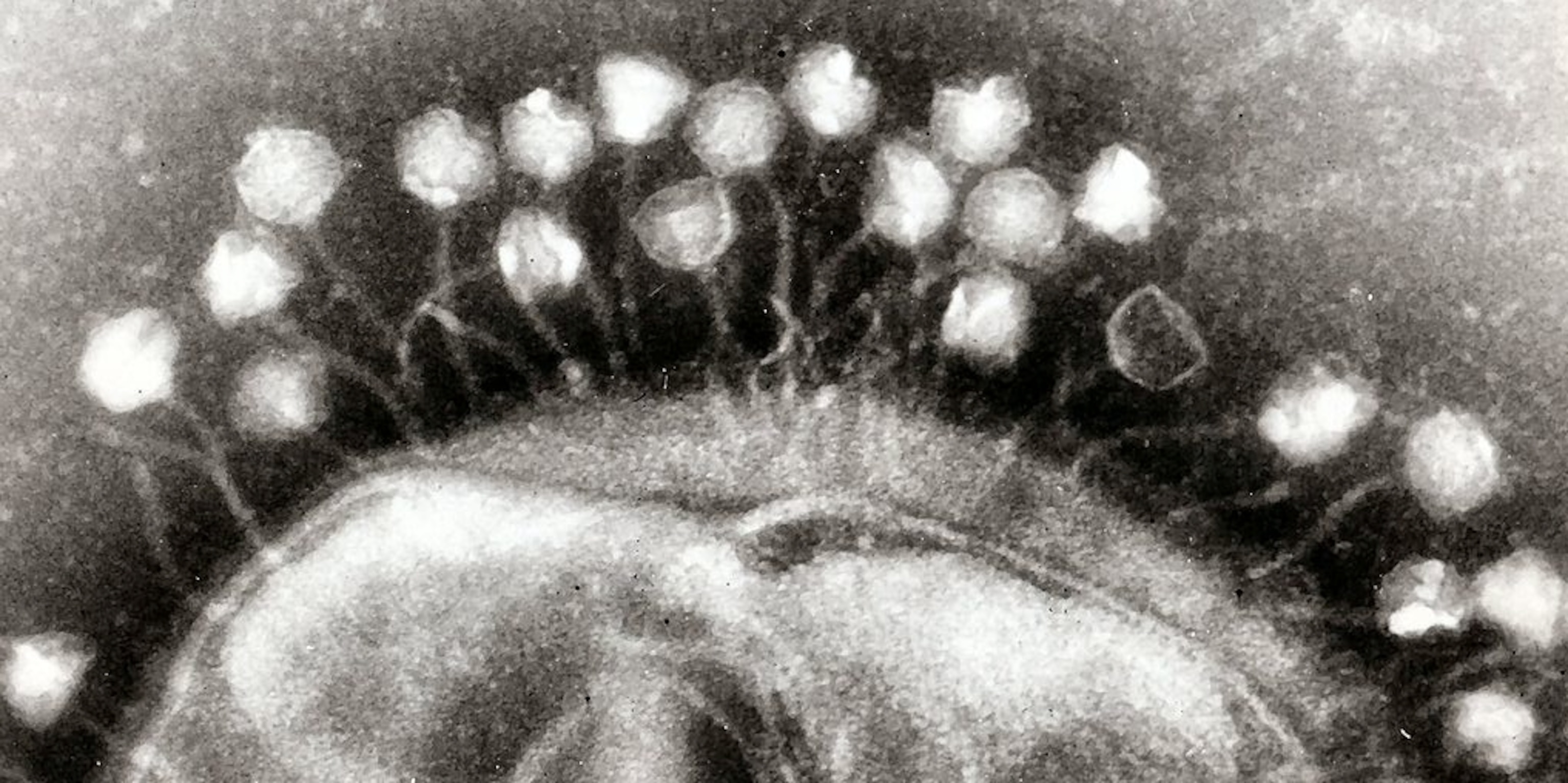
Now comes news of the most bizarre counterweapon I’ve ever heard of in a virus–and a serious challenge to fans of the higher-lower dichotomy. In Nature today, scientists at Tufts University describe their discovery of a virus with its own immune system.
The scientists, led by Andrew Camilli, stumbled across the virus while studying the bacteria that causes cholera, known as Vibrio cholerae. Scientists have long known that V. cholerae gets infected by viruses. In fact, there’s some evidence suggesting that these viruses can bring cholera outbreaks to a halt. As the V. cholerae hosts multiply, their viruses multiply even faster, until they send the bacteria’s population crashing down. Camilli and his colleagues set out to survey these viruses, to see how many species were making life hard for the bacteria.
They revisited a decade of cholera outbreaks by analyzing frozen stool that Bangladeshi doctors had stored from patients between 2001 and 2010. In those samples, they came across 15 different cholera-attacking viruses–12 of which were new to science. Fourteen of those 15 viruses came and went over the decade-long period, causing outbreaks among their bacteria hosts before disappearing.
But one virus–dubbed ICP1–was ominpresent.
Kimberly Seed, a postdoctoral fellow in Camilli’s lab, started sequencing the genes of ICP1 to look for the source its special strength. She found something none of them expected: a full-blown set of CRISPR genes.
Why would a virus carry a set of genes that bacteria use to destroy viruses? To use them against bacteria, it turns out. The ICP1 virus carries barcodes in its CRISPR genes that match pieces of its host’s own DNA. In particular, they match bits of DNA from a set of genes in V. cholerae that interfere with the production of new viruses. In a series of experiments, the scientists demonstrated that the ICP1 virus uses its CRISPR immune system to attack its host’s virus-attacking genes.
In one particularly cool experiment, the scientists engineered the V. cholerae hosts so that their DNA no longer match the virus’s attack molecules. The mutant bacteria managed to destroy most of the viruses. But over time, a few of the viruses somehow managed to acquire bits of DNA from the host and insert them into their CRISPR genes. The viruses regained the ability to shut down their host’s defenses and were able to invade successfully again.
In other words, the viruses had learned something about their enemy.
The ICP1 virus didn’t evolve its own CRISPR genes on its own, the scientists conclude. It stole them. Viruses sometimes pick up host genes and incorporate them into their own genome. The CRISPR genes in ICP1 most closely match those of the bacteria that cause bubonic plague. Long ago, it seems, the ancestors of ICP1 grabbed an immune system from that lineage of bacteria. Later, they turned this bacterial immune system against bacteria.
There are lots of very practical reasons to study immunity in the microbial world–reasons that sometimes only become clear in hindsight. As I wrote in my book Microcosm, the discovery of restriction enzymes in the 1960s made modern biotechnology possible. Scientists used the enzymes to cut and paste genes from one organism to another, creating microbial factories such as E. coli that makes human insulin. Last year, scientists reported that they had harnessed CRISPR genes to create a far more powerful way to edit DNA.
The discovery of a virus with an immune system could open up still other doors. It might be possible, for example, to use viruses to fight bacterial infections. In 2008, Camilli and his colleagues showed that viruses can prevent mice from getting sick with cholera, presumably by killing off the microbes. The mice were not harmed by the viruses, because they are adapted to infect bacteria, not animals.
With an adaptive immune system, these viruses might be able to learn new tricks to overcome any new defenses the bacteria evolve. And just because Camilli and his colleagues first discovered a virus with an immune system in V. cholerae doesn’t mean that there aren’t more of them out there. Indeed, some preliminary database searches hint that they are. Scientists might be able to harness CRISPR-equipped viruses to treat other diseases.
But these practical benefits will take time to emerge, if they ever do. Right now, we can enjoy the brain-stretching experience of looking out at the oceans, the forests, and even in our own mouths, and contemplate the existence of viruses that can learn something about their world.
Lower life indeed.
(For more about viruses, see my book A Planet of Viruses.)