
The Silence of the Crickets, The Silence of the Crickets
In 2003, Marlene Zuk travelled to the Hawaiian island of Kauai and heard something very strange—nothing. A disquieting quiet. An absence of chirping. A silence of the crickets.
Zuk had been studying crickets in Kauai since 1991, back when the insects were both noisy and plentiful. But every time she went back, she heard fewer and fewer of them. In 2001, she heard a single calling male. By 2003, the silence was complete.
The crickets hadn’t disappeared. Zuk would go for nighttime walks and see multitudes of the insects in the light of her headlamp. If anything, there were more of them than before. They just weren’t calling out. When she dissected them, Zuk found out why.
Male crickets call with two structures on the backs of their wings—a vein with several evenly spaced teeth (the file) and a raised ridge (the scraper). When the cricket rubs these together, the effect is like running your nail along the teeth of a comb—you get a thrrrrrrrrrrrp sound. But on all the silent Kauai crickets, the file was growing at a weird angle and had all but disappeared. Their wings were flat.
This change hobbled their courtship songs, but likely saved their lives. In the 1990s, Zuk’s team discovered that the crickets were targeted by a parasitic fly, whose larvae burrow inside them and devour them alive. The flies finds the crickets by listening out for their songs and they’re so effective that, in the early 90s, they had parasitised a third of the males. In 2002, the cricket population had fallen dramatically, and Zuk thought that they were done for.
But the silent males escaped the attention of the fly. As they bred and spread, they carried the flatwing mutation with them. By 2003, the cricket population had rebounded. And in fewer than 20 generations, they had gone from almost all-singing to almost all-silent. The crickets have become a classic textbook example of rapid evolution.
Then, a few years later, the team found that exactly the same thing had happened on the neighbouring island of Oahu! In 2005, for the first time, they found four flatwing males on the island. By 2007, half the males were flatwings.
At first, they thought that the flatwing mutation arose once on Kauai before spreading to Oahu. That made sense: with just 70 miles between the islands, it seemed possible—likely, even—that boats or strong winds carried the flatwing males across to Oahu. When they arrived, they bred with the locals, and their beneficial mutation spread.
But that’s not what happened.
In a new study, Sonia Pascoal from the University of St Andrews has found that this case of evolutionary déjà entendu is actually an example of convergence. The two populations of crickets, threatened by the same eavesdropping parasite, independently evolved similar flattened wings, at pretty much the same time, in just a handful of years.
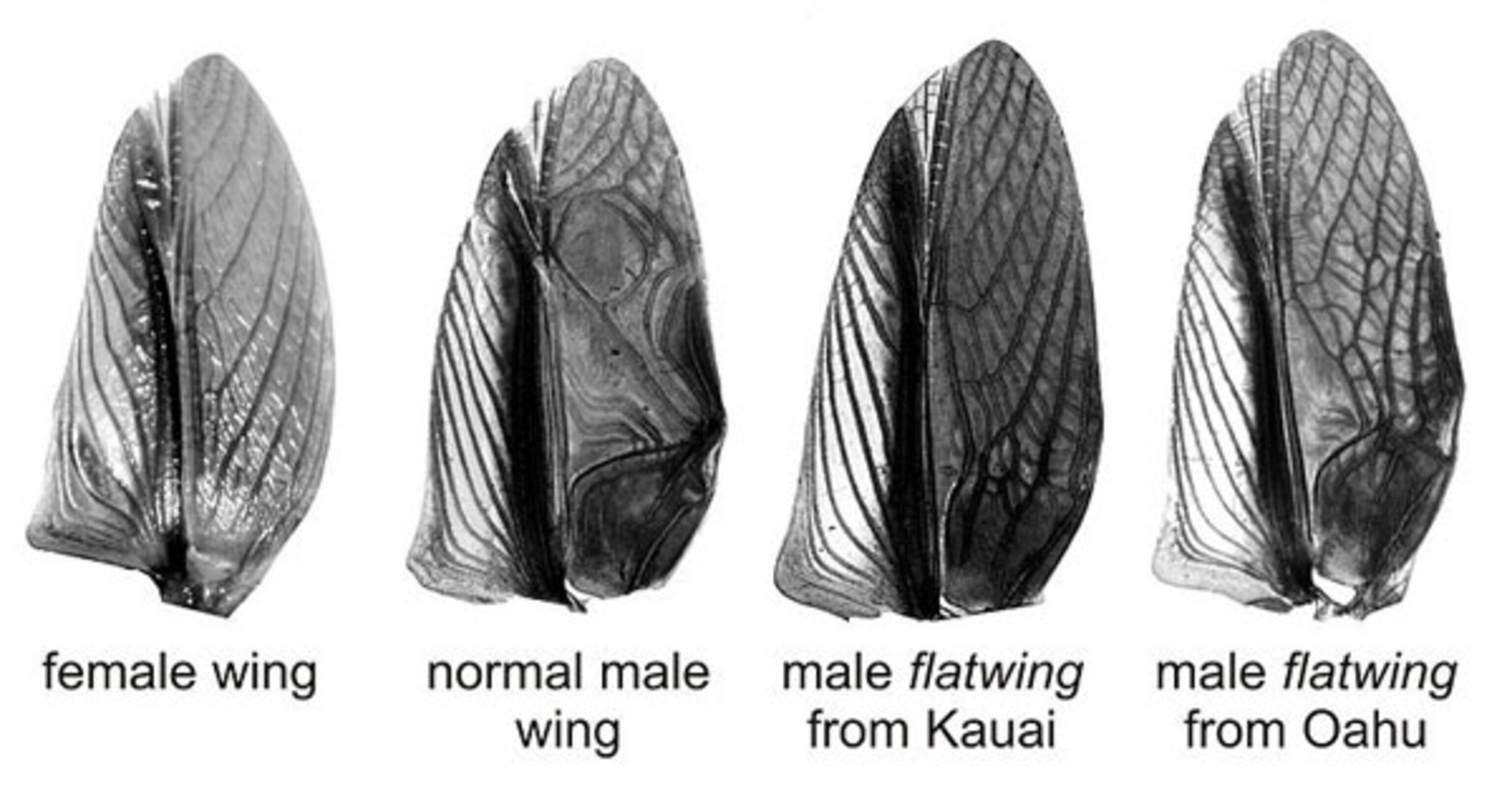
Pascoal’s first clue was that the wings of the silent Kauai crickets look different on those of the silent Oahu ones. You can even tell the two groups apart by eye.
Genetic tests revealed even bigger differences. On both islands, the flatwings are caused by a mutation on a single gene, somewhere on the X chromosome. But both mutations arose independently!
Pascoal’s team looked for genetic markers that flank the flatwing mutation and are inherited together with it. They found more than 7,000 of these, but only 22 were common to both populations. This strongly suggests that the two flatwing mutations arose independently of one another. They seem to have arisen on different versions of the X chromosome. They may even have arisen on different genes or on different parts of the same gene.
“It was quite a surprise!” says Nathan Bailey who led the new study (which Zuk is also part of). “There is solid evidence that evolution can act in the proverbial blink of an eye, but the bulk of this comes from laboratory studies where it is much easier to control conditions. What’s unique about these crickets is the nearly simultaneous appearance of the mutations on two islands.”
The team still have to identify the mutations (or gene) responsible for the flat wings. They also want to know why they arose and what they do. Did the two populations have different starting conditions, that influenced the mutations they eventually gained? Is there a hotspot in the cricket genome where mutations that shape the wings can easily emerge? And do the mutations lead to flat wings in the same way?
The answers will come in time. Just as Zuk’s discovery of the silent crickets gave us a great example of rapid evolution to study, this new discovery provides an excellent opportunity to look at convergent evolution in its earliest stages.
“Many studies that examine convergent evolution are faced with the difficulty that the appearance of mutations that cause similar adaptations in different populations may have occurred very long ago,” says Bailey. “That makes it difficult to tell whether traits with similar functions were derived independently, or whether they share a common ancestry.” I wrote about one such example last week: scientists only recently realised that large, flightless birds like ostriches, emus and rheas evolved their grounded, giant bodies independently of one another.
But on Kauai and Oahu, Zuk and her colleagues have found an example of convergent evolution, happening in real-time. “It’s an extraordinary opportunity,” says Bailey.
Reference: Pascoal, Cezard, Eik-Nes, Gharbi, Majewska, Payne, Ritchie, Zuk & Bailey. 2014. Rapid Convergent Evolution in Wild Crickets. Current Biology. http://dx.doi.org/10.1016/j.cub.2014.04.053