
Contaminomics: Why Some Microbiome Studies May Be Wrong
You’ve got a group of people with a mysterious disease, and you suspect that some microbe might be responsible. You collect blood and tissue samples, you extract the DNA from them using a commonly used kit of chemicals, and you sequence the lot. Eureka! You find that every patient has the same microbe—let’s say Bradyrhizobium, or Brady for short. Congratulations, you have discovered the cause of Disease X.
Don’t celebrate yet.
You run the exact same procedure on nothing more than a tube of sterile water and… you find Brady. The microbe wasn’t in your patients. It was in the chemical reagents you used in your experiments. It’s not the cause of Disease X; it’s a contaminant.
Versions of this story could be playing out in dozens of labs around the world. A team of scientists led by Susannah Salter and Alan Walker at the Wellcome Trust Sanger Institute has shown that DNA extraction kits, and other lab reagents commonly used in microbe studies, are almost always contaminated by low levels of microbial DNA.
Bradyrhizobium is a common culprit, but the team have identified a list of around 100 microbes whose DNA regularly turn up when sequencing supposedly “blank” tubes of water. Most of them live in soil and water. Some come from human skin. This cabal of contaminants, which I’m going to call “the Brady Bunch”, poses a problem for studies of microbe communities, or microbiomes. It raises the haunting possibility that many published results in the field are just wrong.

Salter and Walker first noticed the problem in one of their own studies. Their colleagues had been studying a group of 20 infants, and had swabbed the backs of their noses every month for two years. When Salter’s group sequenced these samples, they found the microbes from the infants’ first months of life were distinct from those that came later. But they soon learned that they had used different DNA extraction kits for the early samples and the later ones, so that both sets were contaminated by different Brady Bunch microbes. When the team excluded these contaminants from their results, the pattern they found also disappeared.
By coincidence, Nick Loman from the University of Birmingham and Michael Cox from Imperial College London had encountered similar troubles. The three groups compared notes and decided to work out the extent of the problem.
Working independently, they sequenced a pure culture containing a single species: Salmonella bongori. Sure enough, that was the only bacterium whose DNA they detected. But when they started diluting the culture, so that there were fewer and fewer microbes in each sample, the results changed dramatically. After five dilutions, S.bongori accounted for just 5 to 30 percent of the results. The rest were contaminants.
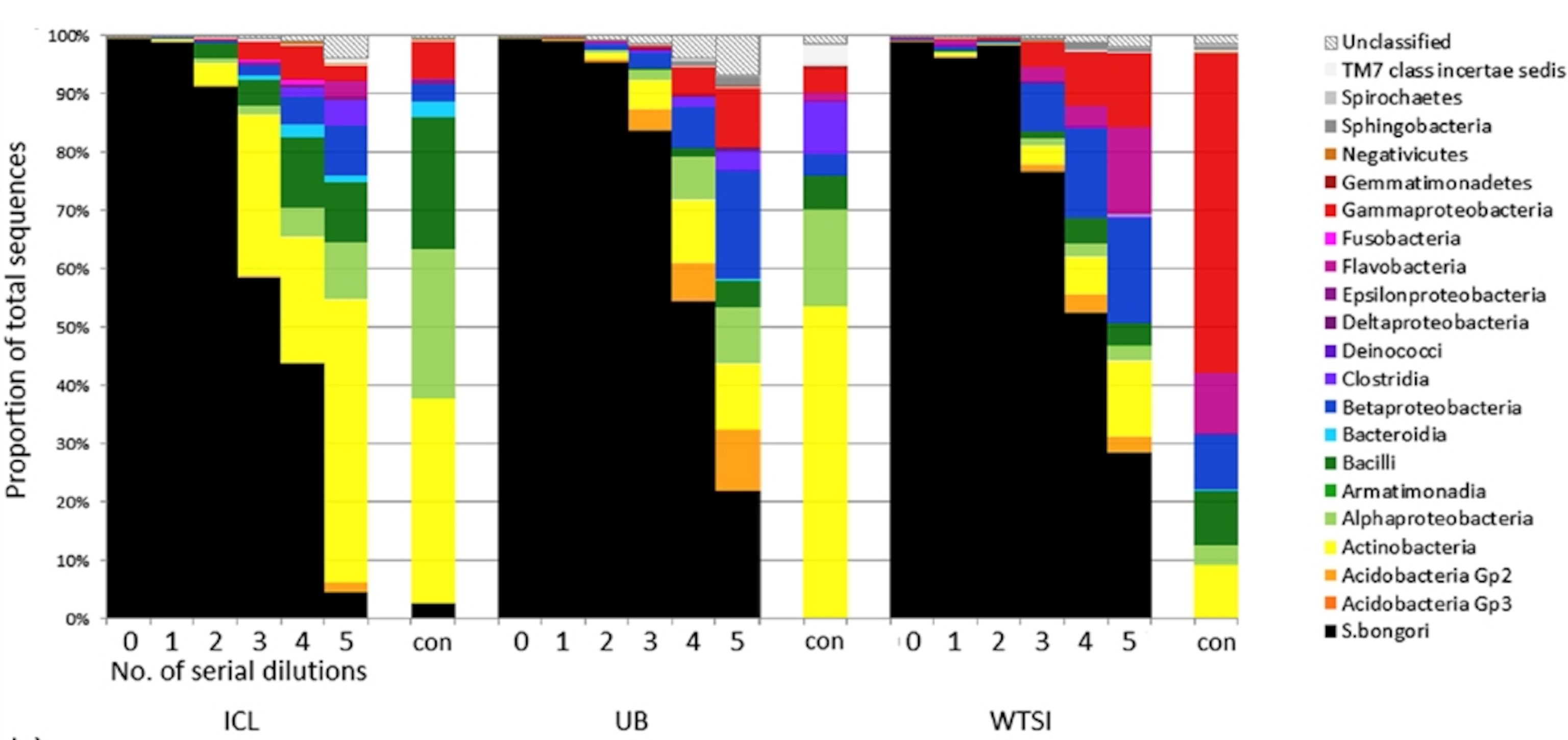
These rogue microbes are inescapable. They vary between different reagents and different labs, but they’re always there to some degree. Loman says that whenever they used DNA extraction kits on tubes of pure water, they almost always get some microbial sequences.
No one knows how many existing studies have been affected by this problem, but the team have highlighted 20 papers that warrant a second look. All of these reported low levels of Brady Bunch microbes. Some were simply cataloguing the microbiomes of habitats like human eyes, earthworm kidneys, and mosquito guts. Others found suspicious species in very unexpected places, including extreme habitats like the upper atmosphere or lakes beneath Antarctica, or places that are thought to be sterile, like the brain or the surfaces of spacecraft.
And some of the highlighted papers found associations between unusual microbes and human diseases. One group reported that the soil bacterium Methylobacteriumsoil bacterium Methylobacterium was more common in breast cancer tissues than in healthy samples, while Sphingomonas was less common. (Delphine Lee, who led the study, says her team used the same DNA kits to compare cancerous and healthy tissues from the same patients, so it’s unlikely that contaminants would be more common in one sample than the other.)
Another team found a new species of Brady in people with an enigmatic diarrhoeal disease called cord colitis, but not in healthy controls or people with other illnesses. That was certainly unexpected. Cord colitis affects people with blood disorders, who are treated with transplanted stem cells taken from umbilical cords. And Brady is a plant microbe—it colonises roots and provides plants with fertilising nitrogen. Could it really be responsible for a human disease?
“I’m alarmed by that paper,” says Loman. “If you screen a bunch of human sequences, you’ll find Bradyrhizobium popping up more often than not. If you find Brady in all of these cases, and no one has ever seen it before, and you haven’t cultured it, then it seems unlikely that you’ve found a novel pathogen.”
But Michael Meyerson, who led the study, counters that his team paid careful consideration to the risk of contamination, and couldn’t find Bradyrhizobium DNA in any of their reagents or control samples, using a variety of methods. “We cannot yet formally exclude the possibility that the finding of Bradyrhizobium in cord colitis specimens is due to contamination, but we believe that the preponderance of the evidence supports the presence of this bacterium,” he says.
These issues are unlikely to matter for, say, surveys of gut microbes, where hordes of bacteria in the actual samples will drown out any rogue contaminants. But researchers should be especially careful when hunting for a rare disease-causing microbe among a throng of other species, or when analysing samples with very few microbes in them at all. For example, scientists who study ancient DNA from fossils often work with minute amounts of DNA. Contaminants are such a huge issue that two researchers recently wrote a strident letter to Science exhorting the field to “do it right or not at all”.
Contamination can also waste valuable time and money and, when it applies to medical research, create false hope for patients. The most egregious recent example involved a virus called XMRV, which was touted as a possible cause of chronic fatigue syndrome (CFS), after a 2009 paper identified it in samples from CFS patients. After a long saga involving much follow-up work, allegations of misconduct, the retraction of the original paper, and much angst for patients, it is now clear that XMRV was a contaminant.
This is not a new problem, but Loman says that he still gets shocked reactions when he presents his team’s results at conferences. “I think many people are still surprised that if you put nothing into your sequencing pipeline, you come out with something that looks like a well-ordered microbiome,” he says. “If you talk about the old guard, they’ll say that contamination has been a problem since day one. But the generation of scientists who are now furiously engaged in microbiome research needs to relearn the lessons of the past. You’ve got to assume that your results might be explained by a technical factor unless you’ve ruled it out.”
“Ruling it out” involves putting everything, even negative controls like tubes of water, through the same process, involving the same reagents. Scientists also need to compare the species they identify against the Brady Bunch list, and take extra precautions if there’s a strong overlap. And perhaps the best test of all would be to grow the microbes from the samples they are supposedly hiding in.
“The significance of this paper isn’t really about contamination, it’s about how we do science,” says Mick Watson from the University of Edinburgh. “Microbiome research is absolutely fascinating, everyone wants to do it, and [we have] the power to explore the microbiome in far more depth than we could have dreamt of before. But with great power comes great responsibility. You need to know the sources of bias and error in your experiment. If you don’t understand that, you’re going to get it wrong. A significant amount of published microbiome research is bunk because people didn’t understand what they were doing.”
Journalists like me, who cover microbiome studies, should also take note. The 20 papers that the team singled out include several that I’ve recently read while researching my book, and at least one that I’ve reported on before. Are these results valid, or are we telling the world about false alarms?
Reference: Salter, Cox, Turek, Calus, Cookson, Moffatt, Turner, Parkhill, Loman & Walker. Reagent and laboratory contamination can critically impact sequence-based microbiome analyses. BMC Biology. http://www.biomedcentral.com/1741-7007/12/87
Note: Apologies in advance to Jonathan Eisen for “contaminomics”. Don’t hit me.