
Despite Attempted Curbs, Animal Antibiotic Use in US Still Increasing
On Friday, the Food and Drug Administration released a vital set of numbers about the routine use of antibiotics in meat animals. The good news: The data is more granular than it has ever been before. The bad news: That granular data shows that most of the trends in ag antibiotic use are headed in the wrong direction.
If you’re a reader who came over from my old blog at Wired, where I talked about this topic a lot, skip down four paragraphs. If you’re new to reading me since I arrived here at Phenomena, stick around for a minute while I give you a short course. Here we go:
In human medicine, we place restrictions on how we use antibiotics. We give them to people with an illness, not people who are well; we give them for bacterial illnesses, not any other condition; and we give them in large-enough doses, over a long-enough period of time, to make sure they cure the illness, by killing the bacteria causing it. We observe those restrictions because we want to keep the bacteria from adapting to the antibiotic and developing defenses to protect itself—something that could happen if the dose is too small, or the drug isn’t the right one to affect the illness.
When antibiotics are used in agriculture, however, we often don’t follow those restrictions. A small portion of antibiotics sold for agriculture in the United States are used to make sick livestock well; but the vast majority go to make animals put on weight more quickly, a practice called “growth promotion,” or to prevent them from developing illnesses caused by the conditions they are raised in, called prophylaxis or sometimes metaphylaxis. Those two uses have been the subject of ferocious policy debate since the 1970s, because they create exactly the conditions that restrictions on human-use antibiotics aim to prevent: The drugs go into animals that are not sick, and are given in small-enough doses that they do not kill bacteria. Instead, bacteria in the animals’ systems adapt to the drugs, becoming resistant to antibiotics and contributing to the international crisis of antibiotic resistance, which kills potentially 700,000 people worldwide each year.
Final thing to know: After decades of stalemate, the FDA is attempting to control how growth promoters are used in the US. In December 2013, it created a three-year program in which manufacturers of veterinary antibiotics agree to change the wording on the labels of their drugs, ruling them out for growth-promoter use. Since label claims have legal force, once “growth promotion” is removed from the labels, the drugs can’t legally be used in that manner. But the new FDA policy doesn’t cover prophylactic use of antibiotics, and advocacy groups say looseness in labeling will allow the drugs to be used unchanged.
OK, now we’re caught up. On to the news:
The data released Friday, under a law called ADUFA (the Animal Drug User Fee Act) summarizes reports made in 2013 by pharmaceutical manufacturers to the FDA, and represents that agency’s best knowledge of how many antibiotics are being sold for agricultural use. As a program, ADUFA has been around only since 2008, while growth-promoter antibiotic use has existed since the 1950s; so the agency has only a few years of data to report, a pretty short trend line.
The trend line, though, still seems to be going up.
- All food-animal antimicrobial sales (in the US): up 17 percent from 2009, the first set of data, to 2013, the most recent data; and up 1 percent between 2012 and 2013.
- Medically important antimicrobials, that is, drugs that are also important in human medicine: Up 20 percent since 2009 and 3 percent since 2012.
- Not medically important antimicrobials (that is, drugs not used in humans, so resistance would be less worrisome): up 14 percent since 2009, but down 2 percent since 2012.
Overall, there were 14.79 million kilograms (32.6 million pounds, or 16.3 tons) of antibiotics sold for use in animals in the United States in 2013. That’s in comparison to a much smaller amount sold for use in humans. The FDA is prickly about not making such comparisons, arguing that the data sets are not comparable. But to give you an idea: In 2011, the most recent year for which there is data, there were 3.29 million kilograms of human-use drugs sold in the US; in that year, sales for animal use in the US were 13.57 million kilograms, about four times as much.
What drugs are being used in animals? The most common, according to the new data, is tetracyclines, which obviously are used in humans also and at 6.51 million kg make up 44 percent of the domestic total. After that, ionophores, an animal-only drug class that at 4.43 million kg makes up 30 percent of the total. Below those two, there are other drug classes that will be familiar from the medicine cabinet: in descending order, penicillins, macrolides (such as erythromycin), sulfa drugs, lincosamides (such as clindamycin), and aminoglycosides (such as gentamicin).
Some other important points: 99 percent of the drugs sold, whether medically important or not, were sold over the counter, that is, without the involvement of a veterinarian. Meanwhile, only 17 percent of the human-significant drugs, and 11 percent of the animal-only ones, were used solely for treatment of animal illness.
The most important data, I think, is in the five-year trends, because they suggest how difficult a task the FDA faces in trying to reduce agricultural antibiotic use. Take a look at this table. Out of 10 drug categories (which include a group the FDA calls NIR, for “not independently reported” because to do so would reveal proprietary data—if a drug is made by only one manufacturer, for instance), between 2009 and 2013, usage rose in seven of them. Even during 2012 and 2013—after the FDA had declared it would try to persuade industry to cut back on growth promoters, but before it finalized the program to do so—usage rose in four of the 10. And all four of those were human-significant drugs—the ones the FDA most wants to see reduced.
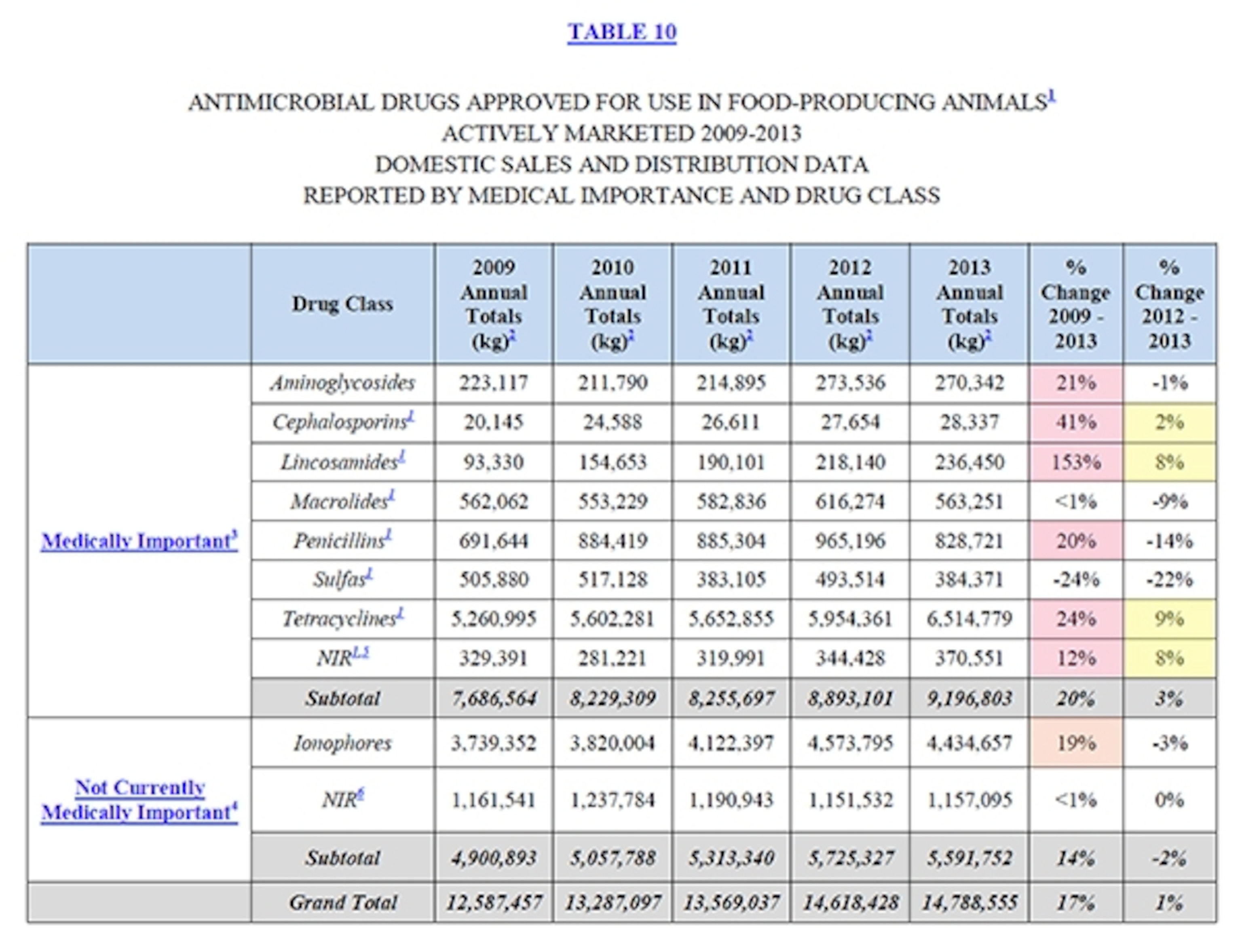
The FDA began making noises in 2010 about introducing restrictions on food-animal antibiotics; so it’s dismaying to see the evidence that, from that point, antibiotic use only went up. The agency’s new program for reducing growth-promoter use went into effect in December 2013. So it’s the next set of data, the ADUFA report for 2014, that should begin to tell us whether it has had a real impact—or whether, as activists fear, it has been treated like a shell game.