Foetal Cells Hide Out in Mum’s Body, But What Do They Do?
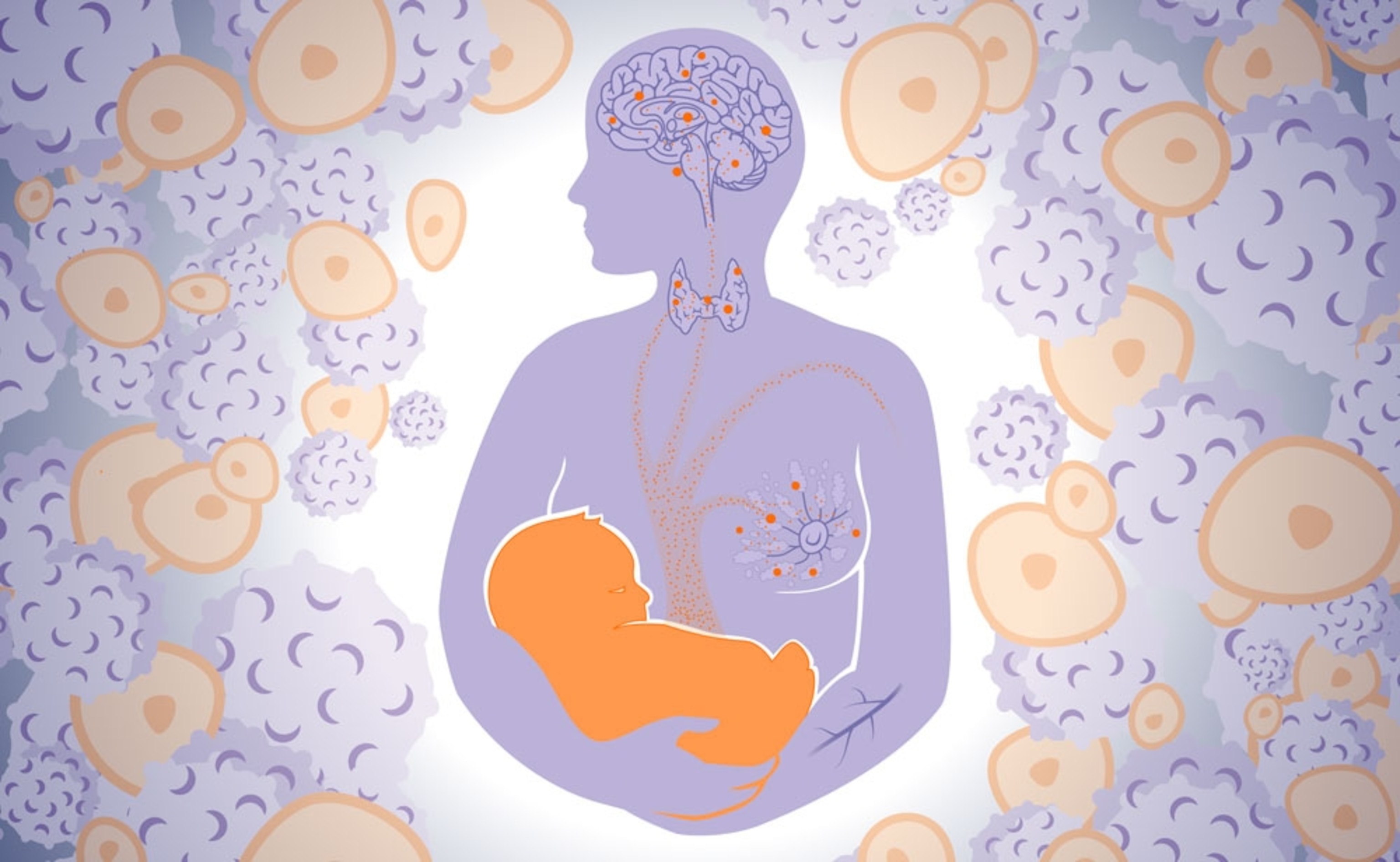
A mother’s children will remain part of her long after they leave her body and enter the world. This isn’t just a saying or a metaphor; it’s biological reality. Every foetus sends some of its own cells into its mother. They cross the placenta, travel through her bloodstream, and lodge in various tissues: brain, thyroid, breast, and more. And then, they stay there. Even after the baby is born, takes its first steps, learns to speak, goes to school, gets a job, and perhaps even becomes a parent itself, some of its cells linger on in its mother.
This phenomenon is called foetal microchimerism, a name that harkens back to the monstrous lion/goat/snake hybrid of Greek mythology. But human chimeras are neither monstrous nor mythical. As fellow Phenom Carl Zimmer wrote in 2013, “scientists are discovering that—to a surprising degree—we contain genetic multitudes”. Twins can pick up cells from their siblings while in the womb. People can end up with several genomes because they arose from two separate fertilised eggs that fused together. And since the 1970s, scientists have found that mothers can harbour their babies’ cells.
These transfers happen during the first trimester of pregnancy, when the placenta hooks up to the mother’s blood supply. The migrant cells are likely to be some type of stem cell: rather than being set in their ways as muscles or skin or neurons, they can transform into many types of tissue. Which type probably depends on where they end up. If they lodge in the breast, for example, they give rise to breast cells. This might explain why the cells are so long-lived. Although many are cleared out of the mother’s body by her immune system, those that infiltrate her tissues could hide out for years, perhaps decades, becoming a genuine part of her body.
But what, if anything, are these cells doing?
In mice, foetal cells accumulate at wounds and injuries, and stimulate the healing process; that might explain why they’ve also been found in healed C-section scars from human mothers. In the breast, thyroid, brain, heart, and skin, they’re sometimes found more frequently in cancerous or diseased tissues, but sometimes more so in healthy, normal tissues. The balance of helpfulness and harmfulness seems to vary from study to study (and since these results are all correlations, it’s possible that the cells have no effect at all). But this picture, though apparently confused and contradictory, makes sense when viewed through the lens of evolutionary biology.
“Mothers and babies have shared interests, but also the potential for conflict because they don’t have completely shared genomes,” explains Amy Boddy from Arizona State University. “It may be optimal for a mother to invest resources in lots of offspring, while for any offspring, it would be optimal to monopolise those resources.”
Such conflicts aren’t deliberate or calculating. They play out through changes in hormone levels, the composition of milk, cries and smiles and tantrums—and perhaps microchimeric cells. In a new paper, Boddy and three other colleagues argue that such cells could provide a way a baby to inadvertently manipulate its mother’s body in ways that benefit it.
Some of those changes, like faster healing, benefit the mother too. Others may not. For example, foetal cells could stimulate the breast to make more milk, either by releasing certain chemical signals or by transforming into glandular cells themselves. That’s good for the baby but perhaps not for the mother, given that milk takes a lot of energy to make—mothers literally dissolve their own bodies to create it. And if the foetal cells start dividing too rapidly in the breast, they might increase the risk of cancer.
Similarly, the thyroid gland produces hormones that control body temperature. If foetal cells integrate there and start dividing, they could ramp up a mother’s body heat, to a degree that benefits her baby but also drains valuable energy. And again, if they divide uncontrollably, they might increase the risk of cancer. Indeed, thyroid cancer is one of the only types that’s more common in women than men, but is not a reproductive organ like the ovaries or breasts.
These subtle conflicts have played out over hundreds of millions of years, and mothers may have well have developed countermeasures. For example, breast tissues might slightly suppress lactation in anticipation that incoming foetal cells would enhance it. “Maybe in the breast, you’d need a certain amount of microchimeric cells to have a good milk supply,” says Athena Aktipis, another co-author on the paper. “These are all speculations but they emerge from taking this evolutionary framework.”
The point is that you’d expect foetal cells to both help and harm a mother, depending on the circumstances. “It’s a game of moves and counter-moves where the foetal system is trying to do more, and the maternal system is trying not to give in to every whim,” says Boddy.
The team are now thinking of ways of testing their hypotheses. It won’t be easy, especially since the ways in which scientists currently test for foetal microchimerism are pretty crude. The most common method is to look for DNA from the Y chromosome in the body of a pregnant woman. Obviously that doesn’t detect cells from female foetuses. It doesn’t even properly detect cells from male foetuses, since the Y chromosome is the smallest of the chromosomes and the most easily lost.
Most studies have also been fairly unambitious. They’ve simply asked: Are these cells there or not? Are there more of them in healthy tissues, or diseased ones with tumours or other problems? That’s a reasonable start but if they are more abundant in diseased tissues, what does that mean? Are they causing disease? Or are they simply flocking to inflamed or cancerous tissues—or wounds for that matter—because more blood rushes to such places?
“Instead of finding whether they’re there or not there, we should be trying to work out what they’re actually doing,” says Melissa Wilson Sayres, one of the co-authors of the new paper. That means collecting the foetal cells and sequencing them to work out which genes they are activating in different parts of the body. The team can then check if these patterns correspond to traits in the mother, like body temperature, or milk quality. “We’re hoping to have a workshop in the spring where we bring experimentalists and clinicians, brainstorm some testable hypotheses, and work out which of these are the most important from a clinical perspective,” Sayres adds.
And then, there’s the matter of cells that travel in the other direction—from the mother to the foetus. What do they do in their new homes? These paths can get even more complicated. It’s possible that the cells from one foetus can travel into its mother, hide out, and then into a sibling during a later pregnancy. “At one point, we started trying to draw family trees, and trying to work out where all the microchimerc cells could be going,” says Aktipis. “It got really messy.”
Reference: Boddy, Fortunato, Sayres, Aktipis. 2015. Fetal microchimerism and maternal health: A review and evolutionary analysis of cooperation and conflict beyond the womb. BioEssays. Http://dx.doi.org/10.1002/bies.201500059