The Forest In Your Mouth
The study of the human microbiome—the booming and much-hyped quest to understand the microbes that share our bodies—began in the mouth. Specifically, it began with dental plaque.
In 1683, Antony van Leeuwenhoek, the first human ever to see bacteria, became the first human ever to see his own bacteria. Untrained as a scholar but insatiably curious, he removed some of the thick plaque at the bottom of his teeth and examined it with his own hand-crafted microscopes. He saw multitudes of living things, “very prettily a-moving”, from spheres that spun like a top to rods that darted through water like fish. Enthralled, he soon started collecting plaque from the local citizenry and finding similar microbes within.
Mouth microbes were largely ignored for the next two centuries, until an American dentist named Joseph Appleton took an interest in them. Compared to microbes in the gut or skin, those in the mouth were easier to collect and less vulnerable to oxgyen. Between the 1920s and 1950s, Appleton and others catalogued these bacteria, and noted that how they were influenced by saliva, food, age, seasons, and diseases. Science historian Funke Sangodeyi notes that these efforts helped to turn dentistry—itself a marginalised part of medicine—into a true science rather than just a technical profession.
In 1999, another dentist scraped some plaque from one David Relman, who then analysed the microbes within it by shredding and sequencing their DNA. That technique is common now, but was groundbreaking then; it took the study of the microbiome to the next level by freeing scientists from the yoke of microscopy and laboratory cultures, and allowing them to more thoroughly identify the microscopic denizens of our bodies. Indeed, even though the mouth was the most well-studied of human microbial habitats, Relman found many new strains and species.
The problem with this sequencing approach is that it’s really good at telling you who‘s there, but not where they are. It’s like studying a city by reading a phone book: comprehensive in a way, but missing a lot of crucial information.
Bacteria are typically a few millionths of a metre long. To them, a human mouth is an entire world. The tongue, teeth, and gums are all very different habitats, each with their own fauna. There are even differences between the microbes below and above the gum line of a single tooth. And within each of these habitats, microbes compete with each other, exchange nutrients, and change the environment around them. To understand this world, and how it might affect our lives and health, we need to take a bacterium’s point of view. We need to know who is where.
Jessica Mark Welch from the Marine Biological Laboratory in Woods Hole and Gary Borisy from the Forsyth Institute have started doing that. From 22 healthy volunteers, they collected scrapings of dental plaque, which they spread onto microscope slides. They painted these samples using differently coloured probe molecules, each designed to recognise a particular groups of bacteria. Collectively, the probes labelled more than 96 percent of the present microbes.
Many researchers have done similar studies with one or two probes at a time, but using 15 at once is really challenging. Mark Welch and Borisy had to ensure that none of them accidentally labelled the wrong group, and that they glowed at comparable intensities. But once they got the technique to work, they started seeing plaque as few others have: in beautiful kaleidoscopic colour.
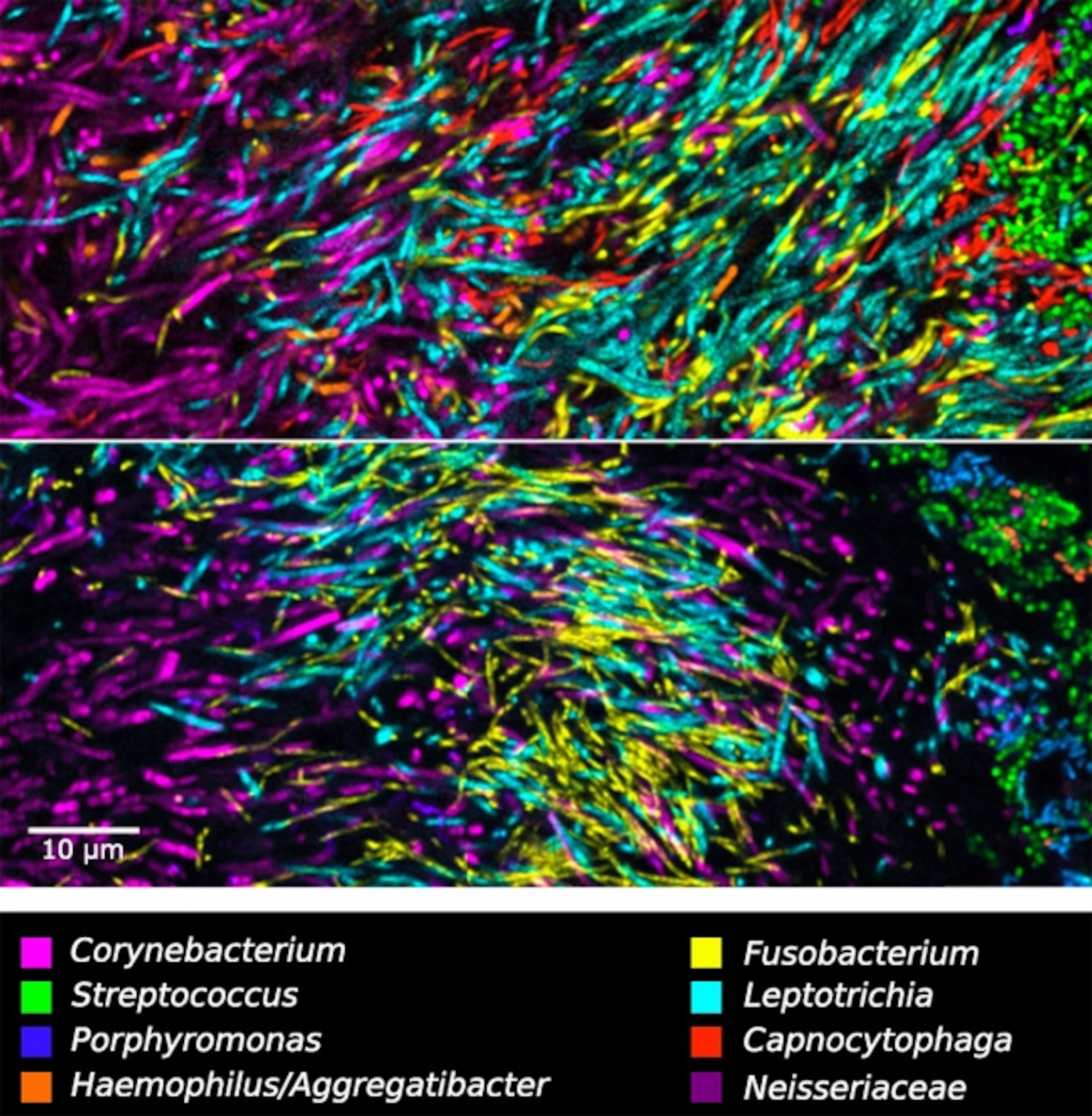
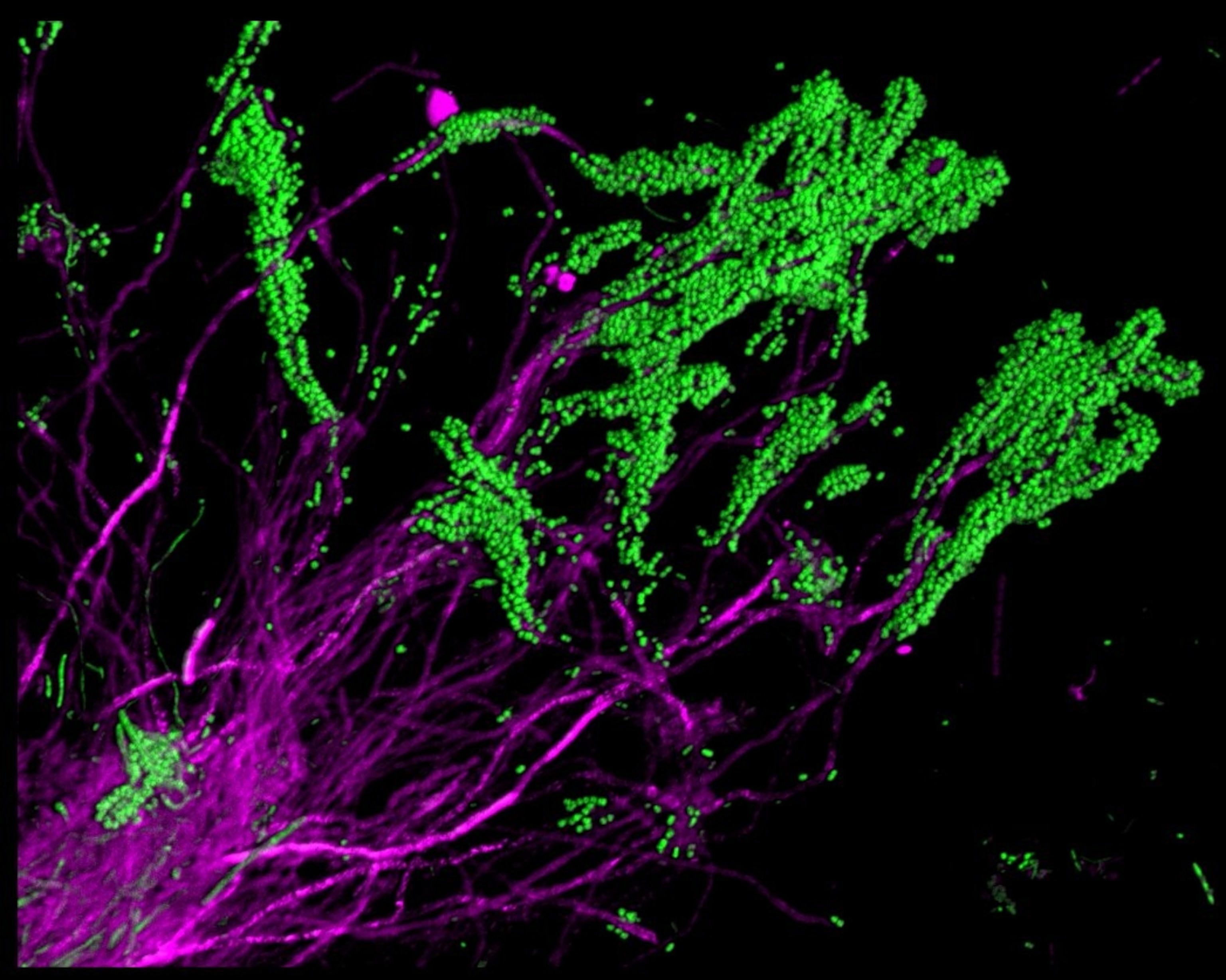
“When we got our first images, we saw these fantastic structures and we were delighted,” says Borisy. There were long magenta filaments of Corynebacterium, sprouting from the same places, and crowned by green Streptococcus spheres. The team called these “hedgehogs”. I see them more like forests, with Corynebacterium as tree trunks and Streptococcus as the canopy. Regardless of metaphor, it’s clear that the bacteria in plaque aren’t just floating about randomly. They’re organised. They’re structured. Each group has its place.
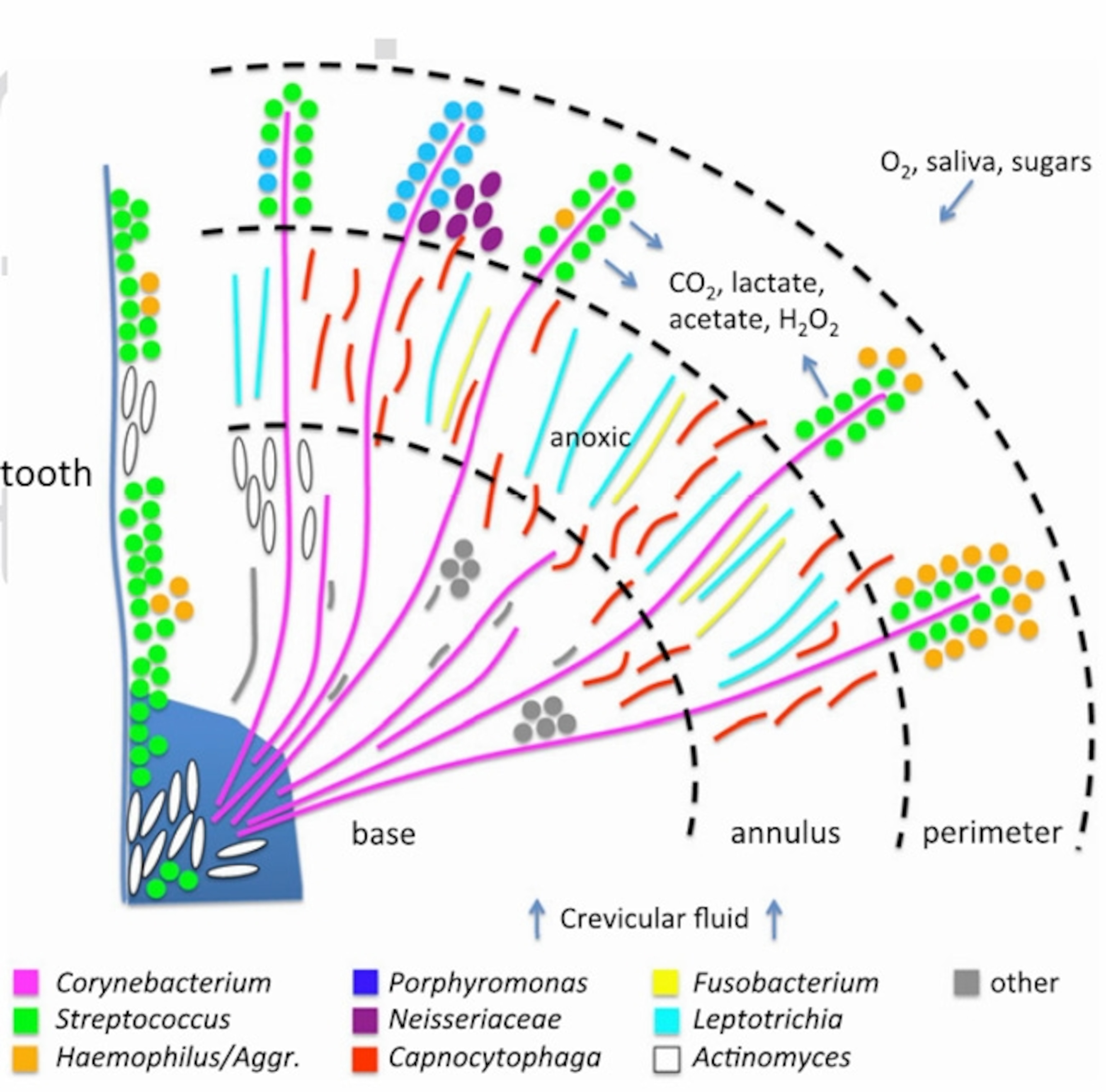
Each species also changes its local environment in ways that others either abhor or exploit. For example, Corynebacterium is the foundational member that cements itself to tooth enamel, grows outwards, and provides a framework for the rest of the community. (It also creates a sturdy reservoir that’s resistant to brushes and mouthwashes, which is why plaque can be so hard to remove.)
In the canopy of the Corynebacterium forest, Streptococcus consumes sugars and oxygen to produce lactate and hydrogen peroxide—substances that many microbes can’t tolerate. But Aggregatibacter can detoxify peroxide and eat lactate, so it also thrives in the canopy.
Streptococcus also releases carbon dioxide, which Capnocytophaga needs to grow, which is why the latter lives in the zone just below the canopy, nestled within the Corynebacterium trunks. This zone, being low in oxygen thanks to Streptococcus, is also home to anaerobic species like Fusobacterium. “This structure was more elaborate and involved than anything we could have imagined,” says Borisy.
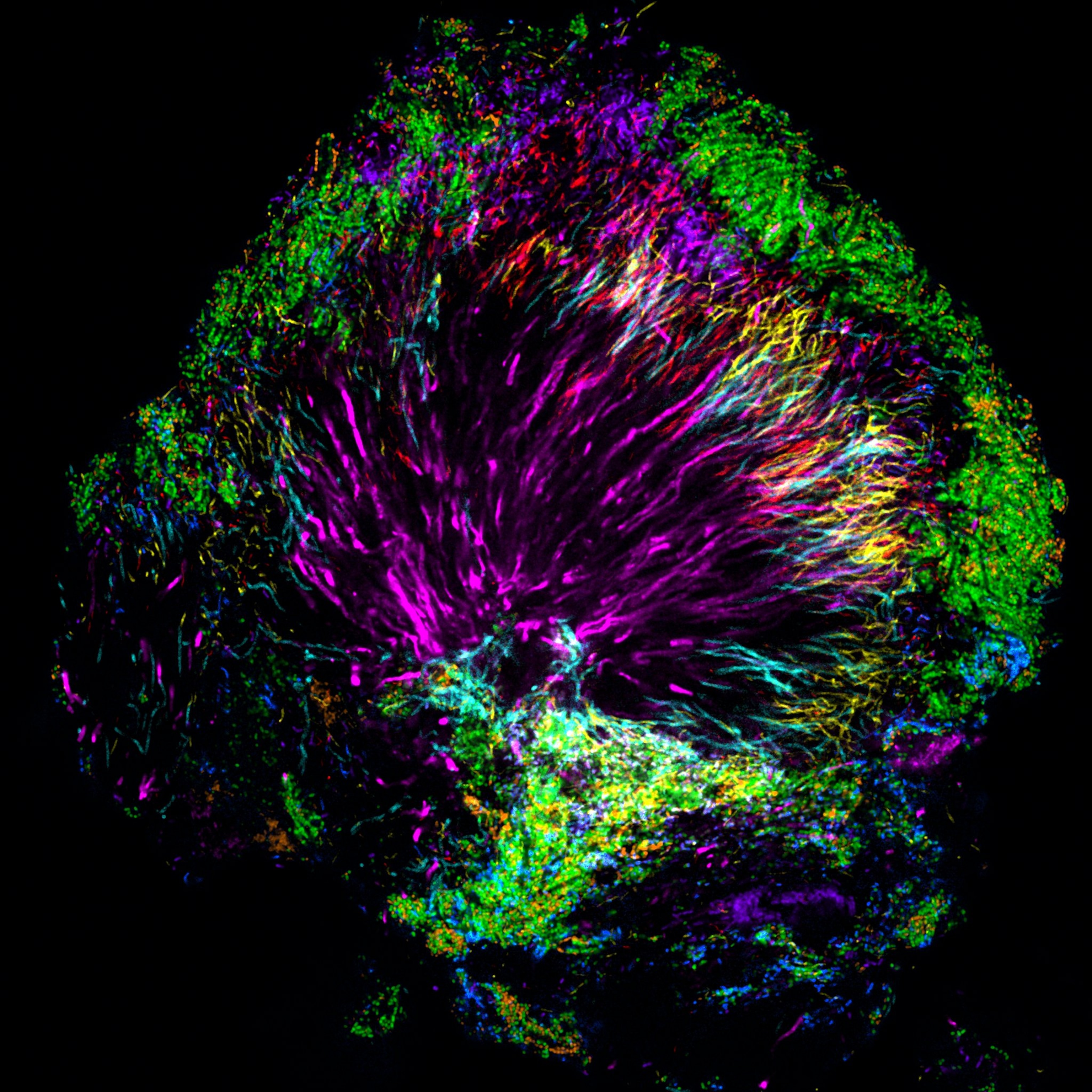
“These results show the very best of what can happen when you look at something well known–plaque–in a new way,” says Michael Fischbach from University of California, San Francisco, who reviewed the new study. “The degree of organization in the community is beyond my wildest dreams. This makes me think plaque is less like a random mixture of bacteria and more like a tissue; organized into sub-structures that probably have specific functions.”
There’s a lot left to learn. For example, how do these forests grow? Clean tooth enamel is first colonised by Streptococcus and Corynebacterium—a slow-growing group—only comes later. How and why does it eventually dominate this microscopic habitat? What happens in cases of gum disease? What happens in other parts of the mouth? Or other parts of the body?
“We’ve begun looking at other sites,” says Borisy. “Plaque was a test bed; the technology is completely general. We could look at other microbiomes in the mouth, elsewhere in the body, in the natural environment, and in the built environment.” But once again, the mouth has led the way.
And once again, crucial discoveries have come from outsiders. Leeuwenhoek was a draper, untrained in science. Appleton was a dentist. Relman started off as an infectious disease specialist, more interested in killing microbes than understanding them. Borisy himself was a biophysicist, who studied the internal skeletons that give cells their structure. “I was having a late-life crisis, and thought I’d start a new line of research,” he says. “And I thought microbes were really interesting.”