They were blinded in accidents. Now, they can see again.
Scientists are making progress on curing blindness with experimental stem cell therapies.
Phil Durst was standing by an industrial dishwasher at Johnny's Restaurant in Homewood, Alabama, telling its owner everything finally sounded alright, when he hit the button that emptied the machine. A hose popped off and blasted him in the face with commercial detergent.
Durst swore, ran to a sink, and began flushing his eyes, but his eyes only felt hotter. He told someone to find lemons to squeeze into his eyes. The acidic juice, he thought, might counter the sodium hydroxide, a basic solution, chewing through his cornea.
After all the juice had been squeezed out, Durst, with difficulty, opened his eyes.
"I couldn't see at all," Durst, 54, says.
Every year, thousands of people are blinded suddenly, like Durst, by injuries to their cornea, the dome-shaped lenses that cap each iris. These injuries are often debilitating. In the weeks after his injury in 2017, all Durst could do was lay in bed in the dark while his wife was at work and sob, not so much because he mourned his vision or his autonomy but because the pain was overpowering.
Doctors often tell patients like Durst they will never see again from their damaged eyes. But stem cell therapies—once the domain of science fiction—have advanced in recent years so rapidly that it's enabling medical breakthroughs.
Recently, for the first time in the US, a treatment has been shown to repair the cornea of people with injuries like Durst. The results came in an experimental trial at Massachusetts Eye and Ear at Harvard Medical School published in Nature Communications last year. Fourteen patients, all of whom had blinding injuries to one eye, received transplants of stem cells to their damaged cornea. At check-ups 18 months later, the surgeon, Ula Jurkunas, associate director of the cornea service at Mass Eye and Ear, found the stem cells had fully repaired the cornea of 10 patients, improving their vision. Durst was the first patient. "I don't know how she did it," Durst said. "It's a work of art."
Researchers are now looking at the study and starting to wonder hopefully: Can science cure other forms of blindness too? "It sets the stage," Jurkunas said of her results, "to go ahead and challenge the system."
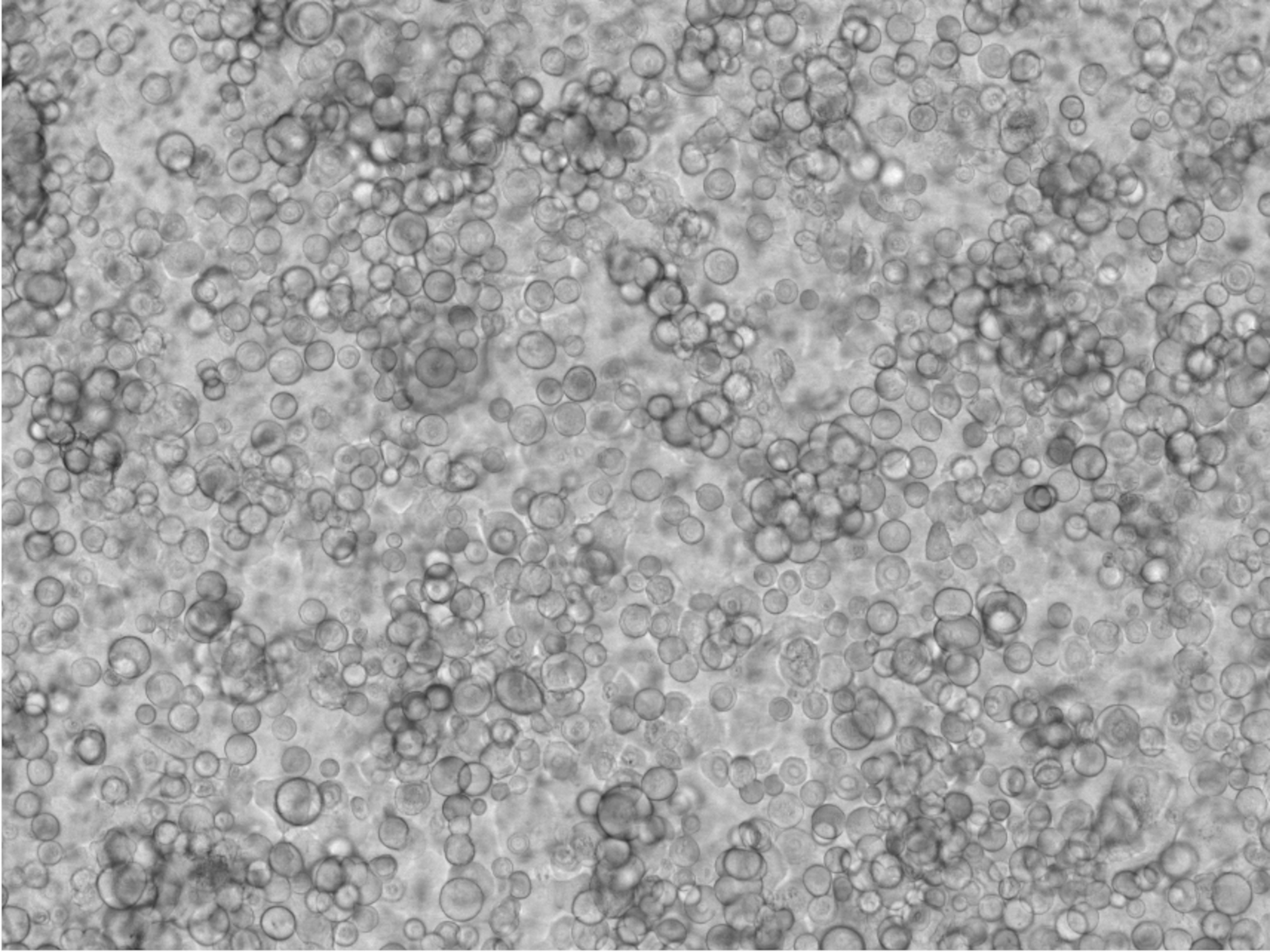
A healthy cornea can constantly repair itself
Cornea are like little dome-shaped windshields that cap each eyeball, and they must remain shiny and clear to focus light deeper into the inner layers of the eye. But even in healthy eyes, cornea are damaged constantly by the mundane violence of daily living: blowing dust, ambient germs, ultraviolet radiation, and the mechanical wear of blinking. We don't typically notice all this damage because our cornea are continuously repaired by limbal epithelial cells, a category of stem cells.
Durst lost nearly all of the limbal epithelial cells in his left eye when the detergent hit him, but he maintained many of them in his right eye. That meant his right eye had the resources to recover some vision—through that eye, he could navigate into the kitchen, slide a meal into the microwave, press the 30-second-start button his wife had struck a strip of red tape to, and when the microwave toned, feed himself.
But his left eye deteriorated, disabling him. The eyeball and eyelid of that eye began growing together without the stem cells, irritating the local nerves, and whenever Durst moved the eye, he set off the nerves. Each time it felt like a carpenter was driving a 10-penny into his skull at the corner of his eye. "Doctors called the episodes cluster headaches. "Until Dr. Jurkunas," he said, "it was just this fact of life."
In 2006, Jurkunas flew to Japan to study a version of the procedure that had been practiced for years in rare cases. Shigeru Kinoshita, professor and chair of Kyoto Prefectural University of Medicine's Department of Frontier Medical Science and Technology for Ophthalmology, had pioneered research on corneal therapies using limbal epithelial cells. But he cultured the stem cells in a solution containing animal products, which the US Food and Drug Administration deemed too dangerous (there's a greater chance mediums made of animal products will infect patients or mutate their genes). Jurkunas brought what she learned from Kinoshita back to the US, creating a solution that resolved the FDA's concerns.
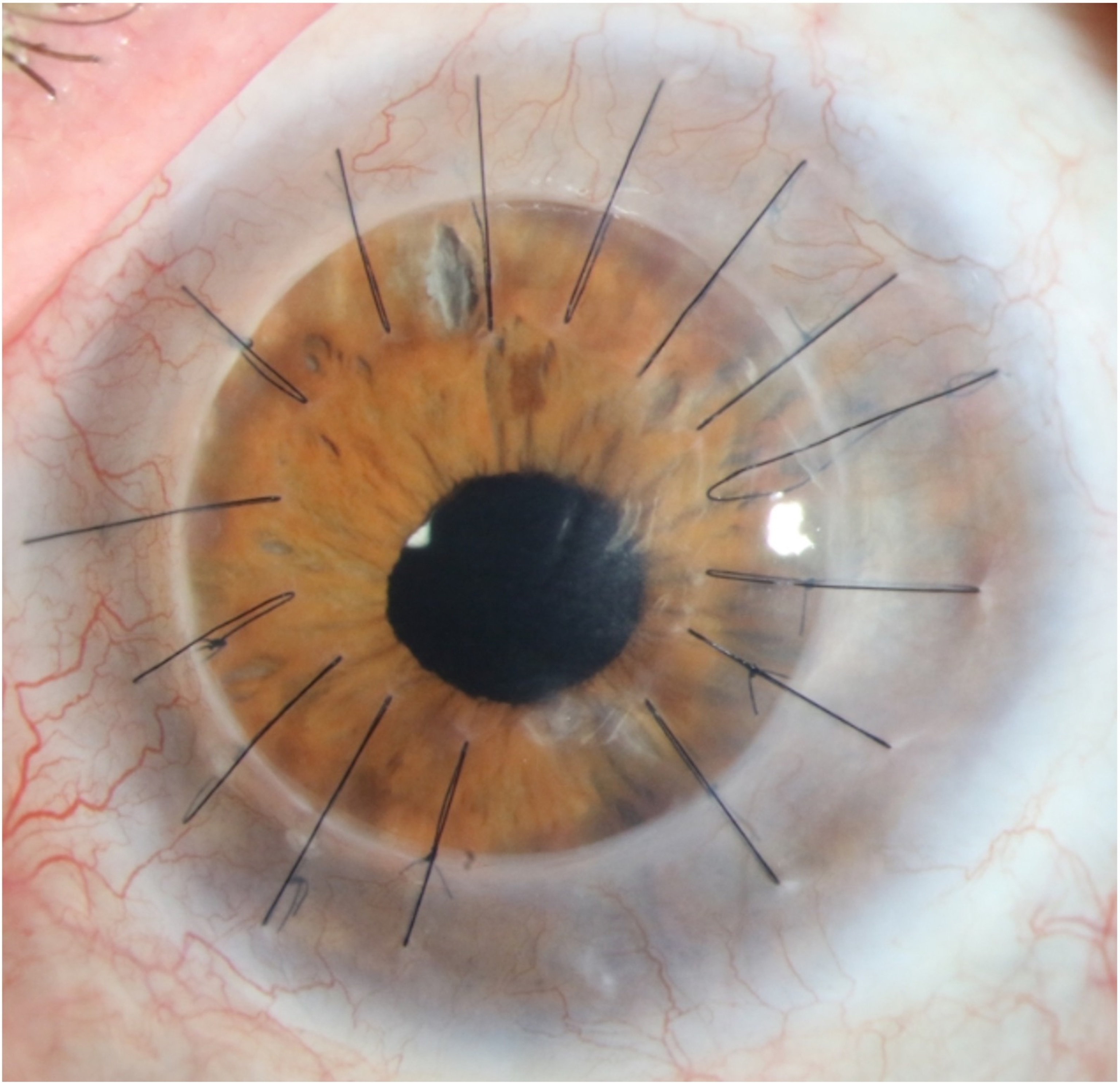
By 2018—after more than a decade of trial and error—Jurkunas had developed her own procedure free of animal products for cultivating the stem cells that meets FDA requirements. It also requires smaller stem-cell samples from patients' healthy eyes and produces larger grafts. The procedure starts with delicately cutting the cells from the cornea of the healthy eye of a patient with a small scalpel called a crescent blade and depositing them into a petri dish with just the right enzymes to destroy unwanted cells. Her team then grows the remaining healthy cells on a strip of plastic over about two weeks, until they form a mat about the size of a penny. From 2018 to 2023, starting with Durst, Jurkunas sewed these little stem-cell mats over the damaged cornea of the 14 patients using sutures as fine as human hair.
After each procedure, the patients returned regularly over about a month in order for Jurkunas and her team to see if the cells were growing well on the eyeball. Jurkunas, who enjoys gardening, compared this final phase of the procedure to transplanting a seedling. "Think of a cell like a plant," she said. "It has to develop roots."
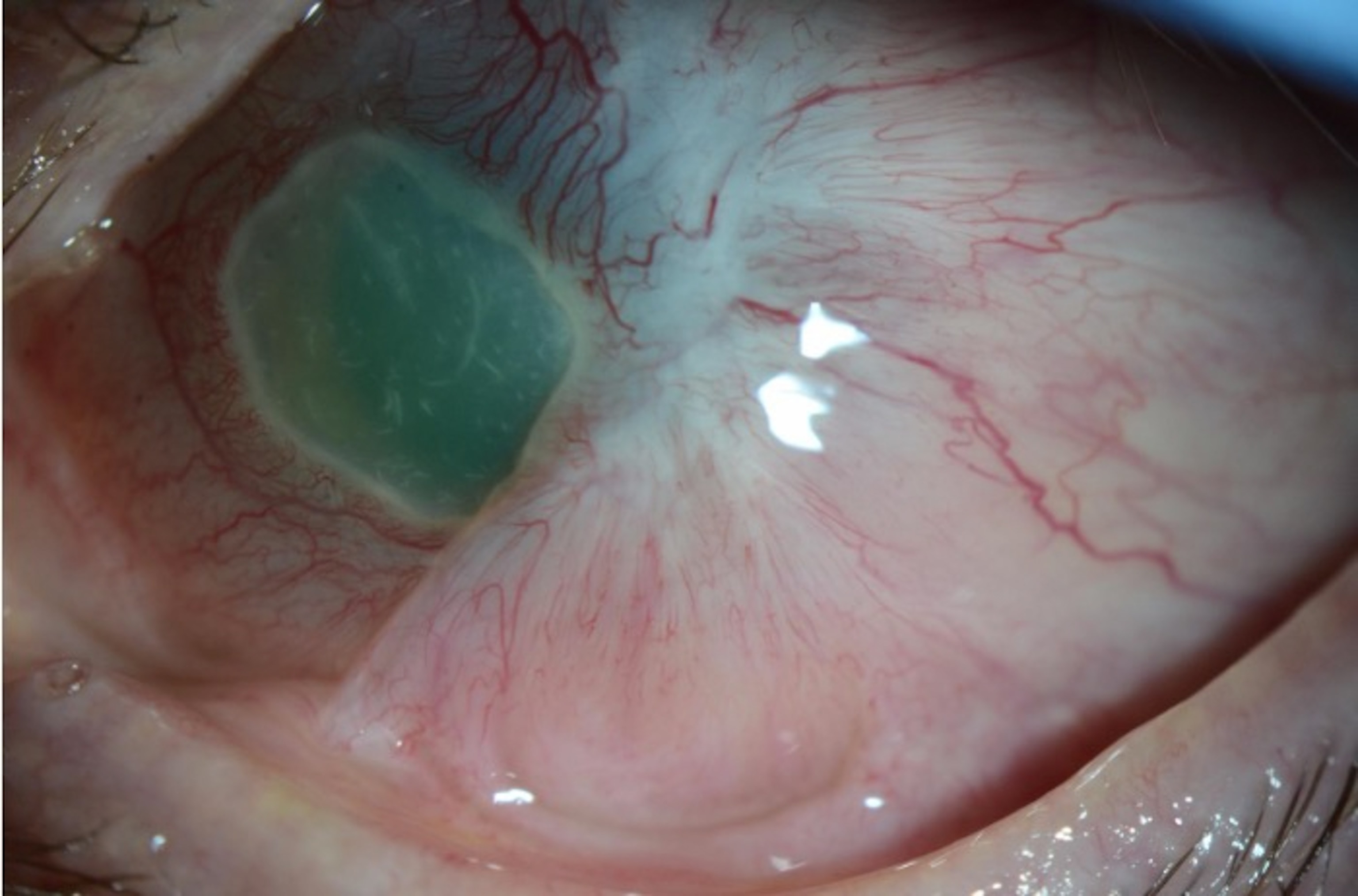
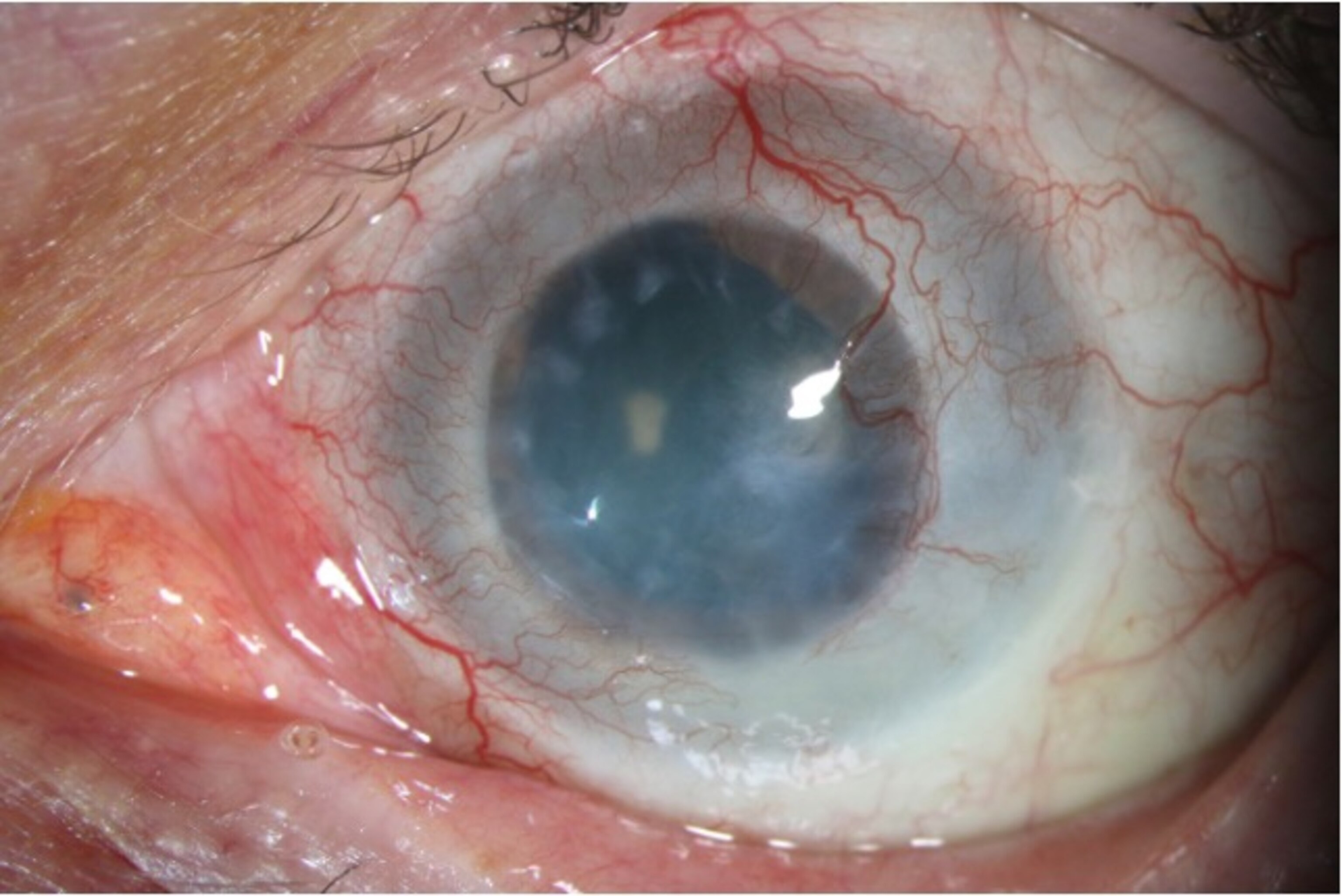
Improved vision—and greatly reduced pain
At a checkup a week later, Durst could see again from his left eye, though not well. Before the surgery, he had been utterly blind from that eye, but now it's as if he sees the world through muddy water, he said in November as he sat in his car. He can see the soda can in his door, and when he holds it six inches from his face, he can read "Fresca." He can also make out the building across the parking lot and the car driving in front of the building. "That's where I am," he said. But, his debilitating pain is gone.
Seventy-five-year-old Carole Cocozza also was a participant in Jurkunas's trial. Cocozza came down with shingles in 2013, and it migrated to her right eye, scaring over the cornea. "I could see light," she said, "nothing else. It was that bad. It was just like looking through wax paper."
Days after surgery, Cocozza said, she could see shadows and shapes from her bad eye. Now the eye is about 80 percent better. She can read a book, if she holds it within a foot of her face. "If something happened to my good eye," she said, "I could take care of myself now."
The procedure blows Durst's mind. It’s like the stuff of Star Trek, he says. During his surgery, not only did doctors restore Durst’s epithelial cells, they also transplanted a new cornea from a deceased donor. (His cornea had been too damaged for the news stem cells to repair.) "It's insane,” he said. “Think of all the things no one could imagine 100 years ago, and now—holy smokes."
Thomas Steinemann, an ophthalmology professor at Case Western Reserve University, does eye surgeries at Metro Health Medical Center in Cleveland, Ohio, a level-one trauma center. In his 36-year career since residency, he’s seen the worst kinds of eye injuries from chemicals, explosions. "It's awful. It's devastating," he said. But now there is hope for these patients, he said; in his lifetime, there will be solutions to their blindness and pain.
"Yes, we're at the early stages of FDA testing," he said of Jurkunas's trials, which have only reached Phase 2 of 3, "but, so far, so good. No safety concerns, and at almost two years, we're talking about a success rate—meaning that the eyes stay healed—of close to 90 percent, which is remarkable."
Sophie Deng, co-chief of the Cornea Division at UCLA's Stein Eye Institute, said it's a big milestone. "These are exciting times," she said. "They did beautiful work to comply with all the regulatory requirements, which is extremely difficult to do."
Could more forms of blindness be treated?
Jurkunas says cornea repairs are the beginning. “This is a concept that can be used for other layers of the eye," she said, like, for instance, the retina, a layer deep inside the eye that converts light into nerve signals. Age-related degeneration of the retina prohibits more than 8 million people globally from focusing on things directly in front of them. Deeper into the eye is the optic nerve, which carries the nerve signals from the retina to the brain, which makes images from them. Glaucoma, a disease that damages the optic nerve, blinds or nearly blinds more than 4 million people worldwide. Research, which Jurkunas is not a part of, is now showing that injecting certain types of stem cells into these diseased tissues can reverse the damage, restoring vision.
Jurkunas is working on licensing the technology so that she can solicit industry funding for phase-three trials, which recruit more patients from across the country and are expensive to run. If trials continue producing safe and effective results, submitting for FDA approval would be next.
If the protocol gets approved, Jurkunas's surgery would be limited to patients who have one good eye from which to harvest donor cells. But many patients do not have a good eye; they are blind in both. To solve for this, Jurkunas is working on cultivating grafts from the limbal epithelial cells of deceased donors, which also comes with a trade-off: She could stockpile the donor cells in a freezer to quickly make grafts when a patient comes to her with injuries to both eyes, but, because the stem cells originate from another person, the immune system would target them for destruction.
Historically, patients who receive foreign transplants take drugs to suppress their immune systems to keep their bodies from killing the transplants, but a suppressed immune system opens a patient up to new dangers. Advances in gene editing—which are progressing in tandem with stem-cell research—are changing this old paradigm. Jurkunas is also trying to find the immune markers of cadaveric limbal epithelial cells so that she can attempt to delete the antigens that trigger the immune system.
Progress elsewhere in medicine suggest this is possible. In the treatment of type 1 diabetes, a pharmaceutical company has demonstrated in clinical trials that it can replace missing insulin-producing cells with stem cells. Another drug company has edited the genes of the insulin-producing cells from a deceased donor and shown, in one patient so far, that they do not produce an adverse immune response. Now researchers are talking about combining the two technologies into a cure.
* * *
Durst used to wake up early to read for two hours before work. He'd read anything: the Wall Street Journal, historical fiction, vampire books.
Even with the repairs to his damaged eye, Durst can't sink into a book anymore. His left eye is still too impaired and the right was always the nondominant one, so it fatigues quickly. "Now I read for purpose," he said, "as opposed to joy." Durst believes that if he had had access to Jurkunas's procedure more quickly, right after the injury, today he'd have more vision out of his left eye. But, as the pain in his eye subsided, his sense of autonomy came back.
He hopes, within the next several years, “this will be in every major eye clinic in the world," he said, "and if it's not, that's a crime."