
Survival of the most cooperative
Can dying coral be nursed back to life by healthy coral? A new theory from a pair of marine biologists studying how the tiny animals work together is offering fresh hope in the fight to save the world’s vanishing reefs.
Rarotonga, the largest island in the archipelago of the Cook Islands, is protected by a coral reef that forms a barricade around the kidney-shaped dollop of terra firma moored in the middle of the South Pacific. If that reef were to die off or disappear—as reefs are, globally, at a disheartening rate—it would spell catastrophe for Rarotonga’s more than 10,000 inhabitants. That partly explains how Anya Brown came to be a regular at a hardware store on the island. The 38-year-old marine ecologist and National Geographic Explorer has been trying to figure out what allows some of the coral in Rarotonga’s turquoise lagoon to survive in increasingly warm conditions. And she needs a steady supply of plastic buckets, PVC pipes, and gloves to do it.
With no research facility on the island, Brown, a professor at the University of California, Davis, has had to improvise to run studies and tend to her growing field nursery. When she started her work in Rarotonga almost two years ago, she and her collaborator, Rachael Bay, 40, another professor at UC Davis, set up an entire ad hoc laboratory on the porch of the Airbnb where they were staying. It was composed of tanks and monitors that controlled water temperatures, and a seawater reservoir that had to be refilled every few hours with five-gallon jugs from the hardware store that they carried to and from the ocean. Inside the tanks, Brown and Bay kept small corals that they collected from around the island, subjecting half to higher temperatures to see what could be gleaned from the resilient ones.
Reefs all around the globe have been in mortal danger for decades—a disaster, considering they harbor an estimated one-quarter of all marine life despite covering only one percent of the ocean floor. Climate change is the main culprit, causing environmental stress and reef disease epidemics. Researchers like Brown and Bay can literally see the struggle: When coral is distressed, it undergoes bleaching, expelling algae living in its tissue and fading in color until it turns white—and dies, if it doesn’t recover. The most recent bleaching event began in 2023 and affected an astonishing 84 percent of reefs worldwide. It won’t be the last. Scientists believe that if the current rate of carbon pollution continues, most of the globe’s coral will die out by the middle of this century.
Brown and Bay are working against the clock to study the survivors—the corals that seem to persist in spite of the disease and stress. “We’re interested in the underlying mechanisms, or underlying reasons, why some corals bleach and some corals don’t,” Brown said. If they can understand coral resilience, they can help preserve these critical species.
But the two scientists are applying two different theoretical systems to the puzzle, a contrast that sits at the heart of modern ecology. Bay is a marine evolutionary biologist, interested in how individual organisms are driven by their struggle for survival. In Bay’s view, natural selection gave rise to corals as they exist now, and the current moment of calamity will force the species to adapt; the resilient ones will persist. To her, the key is helping corals adapt fast enough to keep up with the pace of change. “There’s a lot of talk right now about how to conserve corals under future climates,” Bay said, “and one of the things I think is important is thinking about which corals will survive under future climates—which ones have the right genetic variants to survive warming temperatures.”
Though the collaborators may share an ersatz porch lab and a goal, Brown doesn’t think that the coral will figure it out alone. As a believer in community ecology, she sees species’ lives through their relationships with others. She views coral’s survival as dependent on not just an individual species’ own adaptations but also its documented interconnections with other corals, algae, and even microbes found around reefs. Her intersectional approach is new and unusual in biological sciences but gaining ground (see, for instance, the relatively recent understanding that trees and fungi share resources through underground networks). It means that survival may be less Darwinian—zero-sum, with every species out for itself—and more cooperative.
And on Rarotonga, the pair’s work has started to bear fruit. If they’re successful, they might then be able to draw a road map for helping coral help itself. Proving Brown’s surprising theories, though, depends on cracking open the complex relationships coral relies on. The possible discoveries may radically update our understanding of the role cooperation plays in the natural world.
Corals are strange creatures. They can be as small as a grain of rice or larger than a tree trunk, depending on the species. The fleshy animal itself is an armlike polyp with tentacles ringing its head like a crown. Many corals build armor around themselves, layering calcium carbonate that grows into a multitude of stony shapes: round and squat, filigreed, ruffled, or flat like a stack of spinning plates. And that’s just the coral living in warm water, with access to sunlight—shallow-water coral, the type Brown studies. (Deep-sea coral is a whole other field of study.) But most notably, coral is an organism that throws the whole idea of an organism into question.
Inside its tissue, coral summons and then plays host to tiny, gemlike algae. These algae produce the yellowish green pigment called chlorophyll that absorbs red and blue light from the sun and transforms it into energy to provide their coral host with sugars. In exchange, the algae receive carbon dioxide expelled from the coral animal, nutrients like nitrogen, and protection. This kind of plant-to-animal trade is not unusual. “It is the crux of the cycle of life,” Juli Berwald writes in Life on the Rocks: Building a Future for Coral Reefs. “What is so profound about the alliance between algae and coral is its intimacy.”
Since the relationship was studied and characterized in the mid-1980s, it has become an iconic example of symbiosis, the deep interdependence of living things. Coral hosts the algae alive inside its body, and the algae, once settled, let go of the tiny propellers that give them the ability to swim. The duo almost seem attuned to the needs of the other: Coral produces proteins that act as a sunscreen to protect algae from damaging rays, and algae change the spectrum of pigments they produce, depending on how bright or hot the day is, to temper the negative effects of photosynthesis, like harmful oxygen radicals that injure its cells and stress the coral.
Coral and algae look like—and in critical ways, act like—a single organism. And that relationship ruptures only under the most extreme duress. Bleaching occurrences have become more common as global temperatures rise and oceans become more acidic. Coral can regulate to a certain degree by managing chemical interactions with its symbiotic algae, but only if they remain together.
That’s not the only problem facing coral reefs. Climate change has seeded increasingly furious storms that smash existing reefs to pieces and are followed by currents that damage survivors by swirling the dead rubble across them. Heating ocean waters provide conditions for disease to run rampant, prompting the equivalent of reef pandemics. One, white band disease, attacks the coral’s tissue and kills polyps; stony coral tissue loss disease in the Caribbean creates lethal lesions in the middle of coral skeletons.


If there’s a thin silver lining to coral’s heightened plight, it’s that science has spent more time focusing on the minute organisms themselves—and in doing so, the field has made at least one fascinating discovery. Living coral is covered by a thin layer of mucus that contains millions of bacteria. They’re in the coral’s hard skeleton, and the inch or so of water surrounding the coral too.
Scientists are reckoning with a reality where coral isn’t a two-creature cooperation but a whole consortium of organisms working together to survive. For Brown, that’s vindicating. And complicating. How do you save coral when it requires analyzing not just the intricate relationship between polyps and algae but also the millions of bacteria they live with?
Seven years ago, an accident set Brown down her current path: An outbreak of white band disease had begun creeping toward a nursery that housed 650 tiny corals in the Cayman Islands. She had been working in the nursery for nearly a year, part of her postdoctoral fellowship, when she and a team from the Central Caribbean Marine Institute noticed that disease was starting to affect the specimens. Assiduously collected and planted on underwater PVC frames to serve as an incubator, the nursery samples were meant to replenish reefs that had already been ravaged by white band. As the disease inched closer, she realized the nursery was going to be decimated. By the time the team tried to remove it, there were just too many infected, she said. “We couldn’t really stop it from happening.”
White band is known to begin at the base of a coral structure and creep up like a furry white frost, leaving a denuded skeleton behind. When it first swept through the Caribbean in the 1970s, some reefs lost up to 90 percent of their corals. Scientists couldn’t figure out what caused it. Brown seized the opportunity and organized teams to observe the outbreak in hopes of discovering something new about the disease. They dived every week to study the corals’ condition, taking notes on which ones had been infected, and which had succumbed.
All of the nursery’s frames held coral from the same species, but by fluke, some of the frames held samples collected from a single reef, while others had a mix of corals from different reefs around the island. “It was purely a logistical thing,” Brown said, “and then it ended up being really useful for study design.” When she analyzed the data, she discovered that the “study” had offered a striking revelation.
Corals in the mixed frames—same species, different reefs—survived the disease much better than those that were placed alongside others from the same reef. Brown was startled. “The first time I plotted the data, I had my first ‘oh, whoa’ moment,” she said. “I just couldn’t believe that that was such a strong pattern.”
When she dug deeper, she noticed that the corals most vulnerable to disease fared better in the mixed group than they did surrounded by reef mates. It seemed as though the simple presence of resistant corals could make weaker individuals in the group more resistant. “They were rescued,” she said.
For Brown, the discovery prompted a shift in the way she thought about how individual organisms relate and react to each other. Until that moment, she had mostly considered how different species might symbiotically complement each other, living in a mutually beneficial relationship. That white band outbreak indicated that individuals of the same species might also behave in concert with one another. “I would have expected for the predominant interaction to be competitive,” she said. “And it’s interesting when it’s more facilitative.”


Her study demonstrated the first known connection between intraspecies diversity and disease resilience within coral. A breakthrough in the field, it sparked a question that would shape her work moving forward. If individuals within a species could help each other, she thought, how were they doing it?
During the outbreak, Brown had also taken mucus samples from the coral’s exterior and sent them for bacterial analysis. She compared the data with a similar set that had been gathered before the outbreak and found that certain symbiotic microbes were almost always present on a healthy coral. On a diseased coral, there were markedly fewer.
She hypothesized that corals may lend each other immunity by sharing bacterial communities between individuals. Her theory was built on more than just her own data: Previous research demonstrated that a wide variety of microbes—not just bacteria, but also archaea, fungi, and viruses—could help corals reduce disease.
Microbes’ crucial role in protecting the coral organism was underscored by researchers Forest Rohwer of San Diego State University and Nancy Knowlton of the University of California, San Diego. In their work, they applied new genome sequencing technologies that became more widely available in the 2000s, allowing them to assess microbes that were too numerous to identify by culturing alone. One square centimeter of coral surface contains 100 million microbes, more than 10 times the amount found on human skin, and Rohwer found that more than half the 100 different bacterial species on every genus of coral were, at the time, unknown to science. He also discovered that the relationship between particular bacterial and coral species was specific: Even if corals of the same species were hundreds of miles away from each other, they hosted the same community of bacteria.
Based on Rohwer’s work, scientists began to investigate how they could manipulate microbiomes by adding bacteria and seeing how corals would respond. Microbiologist Raquel Peixoto wondered if it might be possible to actively restore reefs by applying a mixture of beneficial bacteria. “We isolate [the good bacteria] when corals are healthy, and we apply them back when corals are stressed,” said Peixoto, a professor at the King Abdullah University of Science and Technology in Saudi Arabia. “Basically, think of a forest: It’s degraded, burned out. We have to replant trees. We are replanting good bacteria.”
After she applied probiotics to reefs, Peixoto found that a healthy microbiome delayed bleaching—and when corals did bleach, they recovered more quickly.
Peixoto’s work informed Brown’s new hypothesis that microbes might be how corals reach out and help each other. To prove her theory, she’d need to somehow illuminate and quantify this vast and invisible medium that connects corals to each other. It was a task she couldn’t do alone.

Before they were buying buckets at Rarotonga’s hardware store, Brown and Bay were getting to know each other as professors at UC Davis—and realizing that their different approaches, applied together, might give a fuller picture of the characteristics that help corals persist in warming waters. “One of the things that I think is a strength of what Rachael and I are trying to do is mixing our understanding of the coral genome and the microbiome,” Brown said. “We know that a coral is an integrated organism, but we often see studies on one part or one aspect, and not combining across what are really different fields of study.”
Bay and Brown decided to work together in hopes they might come up with a blueprint for coral resilience. Bay suggested pursuing their research in the Cook Islands, where she had done part of her doctoral studies and built relationships with local organizations. They began traveling to the archipelago to understand the state of its reefs. (Their work was supported by the National Geographic Society and Rolex Perpetual Planet Ocean Expeditions, an expansive project sending researchers to study the Earth’s five oceans.) Away from their UC Davis lab, the research required a do-it-yourself mentality and many hours out in the water. They scoured the island looking for study sites, slipping on algae-covered rocks where the sea was shallow and being battered by waves when the wind picked up. It helped that they were both adventurous, enjoyed silly jokes, and loved the movie Pitch Perfect, listening to the soundtrack in their rented truck to hype themselves up for long days snorkeling for fragile coral samples.


Brown and Bay worked closely with Kōrero O Te ‘Ōrau, a Rarotonga NGO that trains young divers to diligently clear the island’s fore reef of crown of thorns starfish, a predator whose numbers have increased as ocean temperatures have risen. Kōrero’s co-founder, Teina Rongo, who has spent almost his entire life in the Cook Islands, told Bay and Brown about his childhood memories of the reef when it was more vibrant and full of life. Three decades ago, fat-lipped giant clams squeezed against one another, and there were octopuses, sea hares, and mats of mussels. Today a giant clam is a rare sight on the reef.
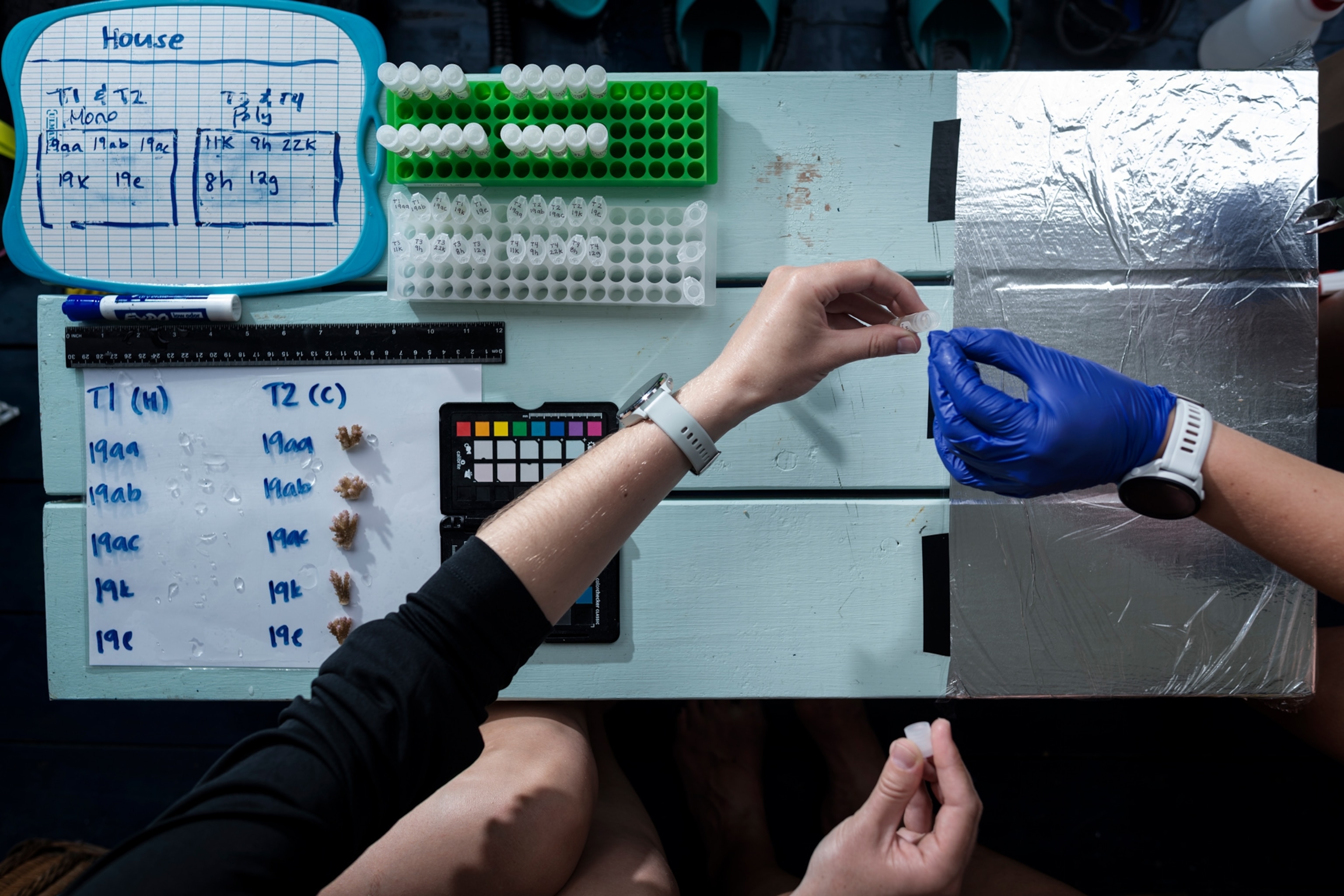
With Rongo’s guidance, Brown and Bay began to develop an understanding of the coral ecosystems and sites across the island. Graduate students helped gather coral samples, specifically the Acropora hyacinthus species, which has multiple fingers growing out of a single branch of rock like a bouquet, making it easy to sample. Then the group planted them in the field nursery to see how mixed colonies, versus a collection from the same reef, fare under the same conditions—a design inspired by Brown’s Cayman Island disease work.
On seven tables made of metal rebar anchored to the seafloor, they zip-tied PVC pipe frames dotted with lumps of coral to monitor growth. Soon after they finished setting up the nursery, in April 2024, a severe heat wave struck. But preliminary data show the corals in the nursery—plucked from sites around the island—proved surprisingly resilient, with 80 percent of those that bleached or partially bleached making a recovery. Though there was no control group to compare against, the experience seemed to corroborate Brown’s approach.
But Bay and Brown wanted a clearer understanding of the factors that determined which corals bleached and which ones didn’t. With the help of their customary hardware store, they set up their temporary porch lab and subjected two batches of corals from their nursery to different temperatures: a baseline, and water warm enough to feel almost like a bath. At the same time, they tracked the conditions of the sites where the porch lab’s sample corals originated. Stitching together the data, they found the corals collected from warmer sites proved more resilient to the relatively hot bathwater-like temps.

Bay’s and Brown’s divergent biological approaches led them in equally divergent—and complementary—directions after that experiment. Bay wondered if the more resilient corals were better acclimated because they had experienced heat before or if it was a sign of evolutionary selection at work. She plans to run genome sequencing to see if she can find particular gene variations associated with heat tolerance.
Brown immediately wanted to explore the microbial communities surrounding the home sites of the most resilient corals. “We are already seeing that there’s genotypic variation in resistance and resilience,” she said. “I think we can start asking more questions about how microbes are underlying some of those characteristics.”
On a bright day last July, Brown and Bay were keeping a close eye on coral pieces collected from Rarotonga’s ocean as the coral sloshed around in a bucket full of seawater. The pair, along with three other researchers from UC Davis, had set up an impromptu laboratory to process samples on a paddleboard that floated gently in the shallow waters. The fragments needed to be driven to the southern part of the island, where they’d be swum out—clutched in someone’s hand—and planted in the nursery, all before dark. It was critical for the corals to stay relaxed, resembling a spattering of tiny asterisks, during their journey. The longer the corals floated in their plastic bin, the more stress they’d endure.
A similar sense of urgency is always present for Bay and Brown as they search for clues to corals’ resilience at the same moment that warming ocean temperatures devastate the species. According to a 2018 study, 14 percent of the world’s corals had already disappeared, with more expected to perish in the coming years. Though the two scientists’ work is ongoing, someday Brown hopes people working on restoration can use their results to ensure the corals with the best chance of survival are given a chance.
As they begin the next phase of their work, they’ve made some critical findings. They’ve noticed that corals better at resisting bleaching seem to have more lipids—insoluble organic compounds, like fat, that are adept at storing energy. And corals that succumb to bleaching but recover quickly have higher concentrations of a molecule called superoxide dismutase, an enzyme that can help protect against oxidation.
But Brown and Bay are still working to articulate the factors that determine resilience. Using collected data, they can now discern whether a hardy coral stays hardy if moved to new places, new circumstances, a new world. It all comes back to the microbes. If corals are planted at a site with reefs known to be resilient, Brown explained, “can they exchange their microbes, for example, for more thermally tolerant microbes?”
That same week in July, the group had also been taking environmental samples from their seven coral collection sites around the island by placing DIY measuring devices—hacked together from plaster of paris glued to light switches from the hardware store—to understand a site’s wave action and the strength of its water current. They also took oversize syringes and swam down to draw water samples next to the study corals.
It’s slow work, and it can feel bleak. Much of the rock wears ghostly fingers of departed coral. Then the scientists would swim around a corner and see a cluster of life: purple and olive green mounds of Porites coral near delicately filigreed yellowish brown brain corals and Acropora humilis, stippled with neon green rings. Clustered towers that branched up, luminous in their strange colors, covered with so many tiny polyps they resembled the skin of lychee fruit.
That afternoon the group picked their way along the craggy reef rocks, looking for more Acropora hyacinthus samples. Brown and Bay roamed farther and farther out, their eyes squinting and necks craning over the shimmering water for the coral’s telltale delicate pink fingers. Bay thought she found a hyacinthus and called Brown over. The coral seemed odd. Each of the branches ended in a stark white, switching to color halfway down the stalk.
“It looks a little pale,” Bay said, frowning.
“Maybe it will bleach and recover really fast!” Brown said.
“Maybe it’s diseased,” Bay said.
They decided to keep looking. By the time the group had gathered about a dozen samples, the clouds had become tinged with pink. Finished collecting, Brown and Bay hauled the buckets onto the back of their truck, rushing to make sure they had everything before their drive.
Twenty minutes later, on the other side of the island, in the dimming light, they swam the corals to their new home. In that moment, the fragments became official study subjects—with Brown waiting to see if they’re the resilient kind. The kind that might help rescue their fellow corals.
